How Do You Test For Oxygen Gas

Ever wondered what makes a campfire roar or why we can breathe and live? It's all thanks to a certain invisible, odorless, and tasteless gas that makes up about 21% of our atmosphere: oxygen! While we rely on it every second, sometimes it's really important to know if it's there, and how much of it. Maybe you're a budding scientist, a curious homeowner checking on your HVAC system, or just someone who loves a good ol' fashioned experiment. Testing for oxygen might sound like something out of a high-tech lab, but it can actually be surprisingly straightforward and, dare we say, even a bit fun!
Why Bother Testing for Oxygen?
You might be thinking, "Oxygen is everywhere! Why would I ever need to test for it?" Well, there are several super-useful reasons. For starters, in many critical situations, knowing the exact concentration of oxygen is vital for safety. Think about astronauts in space, deep-sea divers, or even people undergoing medical treatments – a precise oxygen level is the difference between life and a very, very bad day.
Beyond life-or-death scenarios, testing for oxygen can be helpful in industrial settings. For example, in chemical processes, the presence or absence of oxygen can determine if a reaction will happen or how it will proceed. It can also be used to detect leaks in sealed environments or to ensure the quality of stored goods. Even at home, understanding oxygen levels can be relevant for things like ensuring proper ventilation in enclosed spaces or checking the effectiveness of air purification systems.
Must Read
Plus, let's be honest, there's a certain satisfaction in understanding the world around us, and knowing how to test for something as fundamental as oxygen is pretty cool. It's like having a secret superpower that lets you peek into the invisible world of gases!
The Classic Flame Test: A Fiery Friend
One of the most classic and accessible ways to test for the presence of oxygen is using a simple flame. This method relies on the fact that oxygen is a key ingredient for combustion – it fuels the fire!

Here's how it generally works: You take a small flame, like from a lit match or a candle, and carefully introduce it into the area you suspect contains oxygen. If the flame suddenly becomes brighter, burns more vigorously, or even flares up, it's a strong indicator that there's a higher concentration of oxygen present than in normal air. Imagine blowing on a dying ember and watching it burst back to life – that's essentially what's happening when oxygen is abundant!
The more oxygen available, the happier a flame is!
It's important to be cautious when doing this, of course. You're dealing with fire, so always ensure you're in a safe, well-ventilated area, away from flammable materials. And if you're testing a specific gas sample, make sure it's contained safely. This method is great for a quick, qualitative check – it tells you if there's more oxygen, but not how much more.
The Glowing Splint Test: A Subtle Signal
For a slightly more subdued, but equally effective, test, we turn to the glowing splint. This is a fantastic method, especially when you're dealing with a situation where a full flame might be too much, or when you're trying to differentiate oxygen from other gases.
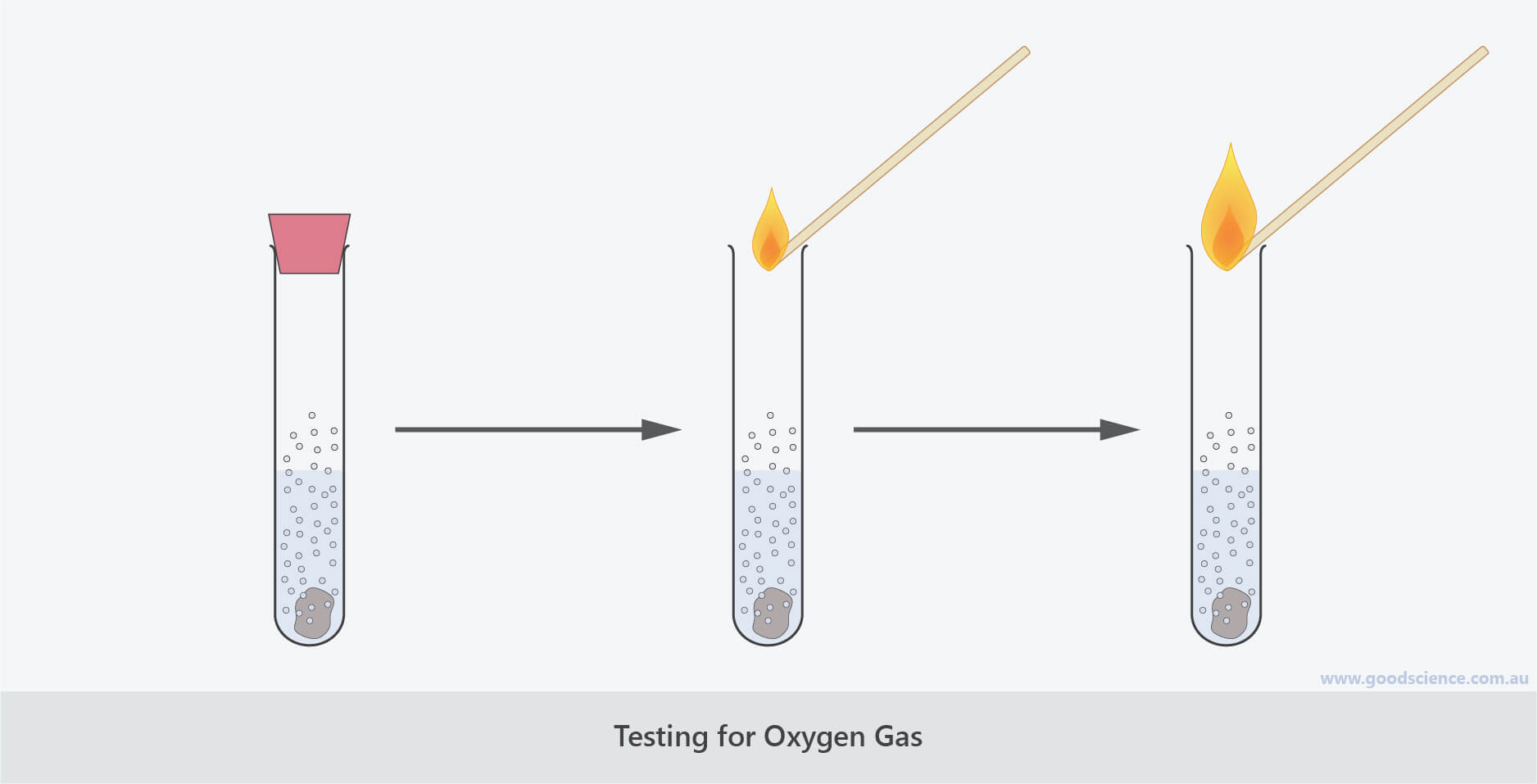
The process involves taking a wooden splint (think of a thin wooden stick), lighting it, letting it burn for a few seconds, and then blowing out the flame. You want to be left with a glowing ember at the tip of the splint. You then carefully insert this glowing splint into the atmosphere or gas you want to test.
If the glowing splint suddenly reignites into a full flame, it's a clear sign of a significant amount of oxygen. The intense presence of oxygen essentially "feeds" the ember, providing enough fuel for it to burst back into visible flames. This is a very common test done in chemistry classes because it's so visually dramatic and easy to interpret. It’s like the splint is saying, "Wow, that's a lot of oxygen! I'm back, baby!"

Again, safety is paramount. Make sure your splint is handled carefully and that you're in an appropriate environment. This test, like the flame test, is primarily qualitative, indicating the presence of a strong oxygen concentration rather than a precise measurement.
Beyond the Flame: More Sophisticated Methods
While the flame and glowing splint tests are fantastic for demonstrations and quick checks, there are many more precise ways to measure oxygen levels, especially when accuracy is critical. These methods move beyond simple observation and into the realm of scientific instrumentation.
One common type of sensor is an electrochemical sensor. These clever devices work by having oxygen molecules react with an electrolyte solution within the sensor. This reaction produces an electrical current that is directly proportional to the amount of oxygen present. The higher the oxygen concentration, the stronger the current. These are widely used in medical devices and safety monitors.

Another sophisticated approach involves paramagnetic sensors. Oxygen is unique among common gases in that it is paramagnetic, meaning it's attracted to a magnetic field. Paramagnetic sensors utilize this property. They typically have a small dumbbell-shaped rotor suspended in a magnetic field. When oxygen enters the sensor, it's drawn towards the magnetic field, causing the rotor to move. The degree of this movement is used to calculate the oxygen concentration.
For even more precise measurements, especially in research or industrial applications, techniques like gas chromatography can be employed. While much more complex, these methods can separate and quantify various gases in a sample, including oxygen, with incredible accuracy.
So, whether you're relighting a campfire, performing a school experiment, or ensuring critical safety standards, understanding how to test for oxygen opens up a fascinating window into the science that surrounds us. It’s a simple element, but its detection can be both elementary and highly advanced!


