Why Does I C E Float On Water

So, picture this: it’s a scorching summer day, and I’m desperately trying to cool down. My go-to move? A massive glass of iced tea. I meticulously fill it, add a generous squeeze of lemon, and then, the pièce de résistance – a handful of ice cubes. As they tumble into the amber liquid, I watch them bobbing around, completely unbothered by the fact that they're surrounded by water. And it always hits me, this little moment of wonder: Why on earth does ice float? It’s water, right? It should sink! It’s like a tiny, frozen rebellion happening in my drink.
I mean, you’d think water would be pretty consistent, wouldn't you? It’s everywhere. We drink it, we bathe in it, we complain about it when it rains. But that simple act of dropping an ice cube into a glass is a daily demonstration of something truly fascinating. It’s not just about keeping our drinks cold; it’s a fundamental property of water that shapes our entire planet. Kind of makes you feel a bit silly for taking it for granted, right?
Let’s be honest, most of us probably don’t spend our free time pondering the molecular structure of H₂O. We’ve got more pressing matters, like figuring out what’s for dinner or deciding which streaming service has the best binge-worthy series. But this whole ice-floating thing? It’s a big deal. It’s the reason we have ice ages, the reason life can exist in frozen lakes, and the reason your fancy cocktail doesn’t just turn into a solid block of frozen disappointment.
Must Read
The Curious Case of the Bobbing Ice Cube
So, what’s the secret behind this aquatic defiance? It all comes down to density. Yeah, I know, density. It sounds like something you’d learn in a stuffy science class and then promptly forget. But stick with me, because density is the key player in our ice-floating drama.
Think of density as how much “stuff” is packed into a certain amount of space. If something is really dense, it means a lot of its mass is squeezed into a small volume. If it’s less dense, the same amount of mass is spread out over a larger volume. Makes sense, right?
Now, here’s where water gets a little weird. Most substances, when they go from a liquid to a solid, get denser. That’s because their molecules pack themselves in tighter and tighter, like a well-organized filing cabinet. But water? Oh no, water likes to do things differently. It’s a bit of a rebel, remember?
Water: The Anti-Social Molecule
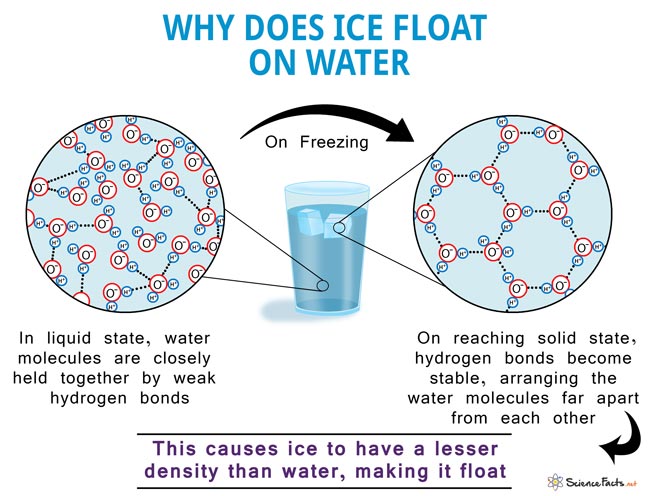
When water freezes, its molecules actually arrange themselves into a specific, crystalline structure. This structure, called a hexagonal lattice, is surprisingly… spacious. Imagine those tightly packed filing cabinets suddenly deciding to become a room with lots of empty chairs. The molecules are still there, but they’re not as close together as they were in the liquid state.
This is because of something called hydrogen bonds. Water molecules (H₂O) have a slightly positive charge on the hydrogen atoms and a slightly negative charge on the oxygen atom. This makes them act like tiny magnets, attracted to each other. In liquid water, these hydrogen bonds are constantly forming, breaking, and reforming. The molecules are jiggling and sliding past each other, able to get pretty close.
But when water freezes, these hydrogen bonds become more stable and lock the molecules into that rigid, open lattice. They have to spread out to accommodate these fixed bonds. It’s like they’re holding hands in a very precise, geometric pattern, and they can’t get any closer.

So, what does this mean for density? Well, if the same number of water molecules take up more space when they freeze, they become less dense. It’s like taking the same amount of Lego bricks and building a sparse, airy castle versus a solid, compact block. The castle uses more space, even with the same number of bricks.
And here’s the kicker: ice is about 9% less dense than liquid water. That’s why it floats! It’s literally lighter for the same volume compared to its liquid counterpart.
The Ripple Effect: Why This Matters (A Lot!)
Okay, so ice floats. Cool. We’ve established that. But why should we care beyond our chilled beverages? Because this seemingly small quirk of water’s behavior has massive implications for life on Earth. Seriously, it’s a planetary-scale game-changer.
Imagine if ice sank. Every time a lake or ocean froze, the ice would sink to the bottom. This would mean that over time, bodies of water would freeze solid from the bottom up. Brrr! No bueno for any aquatic critters trying to survive the winter. Fish, amphibians, all sorts of life would be crushed under the weight of accumulating ice and then permanently frozen.
But because ice floats, it forms a protective layer on the surface of lakes, rivers, and oceans. This layer acts as an insulator. It shields the water below from the frigid air temperatures. Think of it like a cozy blanket for the underwater world.
This means that even in the harshest winter conditions, the water at the bottom of a lake remains liquid. Life can persist. Fish can swim, plants can grow, and ecosystems can survive. It's a literal lifeline for aquatic life.

Ice Ages and Our Climate
And it’s not just about individual lakes. The floating of sea ice plays a crucial role in global ocean currents and climate regulation. When sea ice forms, it leaves behind salt. This makes the remaining unfrozen ocean water denser, causing it to sink. This sinking then drives massive ocean currents, like the thermohaline circulation (try saying that five times fast!).
These currents are like the Earth’s circulatory system, distributing heat around the globe. They help regulate our planet’s climate, making some regions warmer and others cooler. Without floating ice, these crucial currents would be dramatically altered, leading to much more extreme and unpredictable weather patterns.
The formation and melting of ice sheets also played a massive role in past ice ages. As ice accumulated and floated, it altered ocean currents and reflected more sunlight back into space, leading to further cooling. It's a feedback loop that dramatically shaped our planet's history. And guess what? We're still experiencing the aftermath of the last ice age today.
So, the next time you see an ice cube bobbing merrily in your drink, take a moment. It’s not just a frozen bit of water; it’s a tiny, everyday miracle that supports an entire planet.
A Little Molecular Drama
Let’s dig a bit deeper into the molecular level. It’s like peering behind the scenes of a play, watching the actors (molecules) move and interact. In liquid water, the H₂O molecules are constantly bumping into each other, vibrating, and rotating. They're pretty free to roam, like kids in a playground. The hydrogen bonds are there, but they’re fleeting, like brief handshakes.
Now, when you cool water down, the molecules start to slow down. They’re less energetic, like kids starting to get tired at the end of the day. As they reach the freezing point (0°C or 32°F), they’ve lost enough energy to settle into those more ordered, fixed positions. The hydrogen bonds, which are still present, now have enough stability to lock the molecules into that distinctive, six-sided (hexagonal) arrangement.

Think of it like this: imagine you have a bunch of bouncy balls. In liquid water, they’re all bouncing around randomly, filling up the space. When they freeze, they suddenly decide to line up in perfect rows, with a little gap between each ball to hold onto the imaginary string connecting them. That little gap is the key to the lower density.
This open structure is crucial. It’s what makes ice less dense. If water behaved like most other substances, its solid form would be denser than its liquid form, and it would sink. This would be a disaster for aquatic life and global climate.
The Irony of It All
There’s a kind of beautiful irony to it, isn’t there? Water, the very substance that can be so powerful and destructive (think floods and tsunamis), is also incredibly life-sustaining, thanks in part to its tendency to be a bit… aloof when it freezes. It creates a barrier, a protection, rather than a crushing weight.
It’s like the universe has this amazing sense of humor. The very thing that could freeze the planet solid actually provides a way for life to survive those freezes. It’s a subtle, elegant solution that we often overlook because it’s just… there.
And think about it from a human perspective. We use ice for everything from chilling our drinks to preserving food to making snow cones. We carve it, we shape it, we build with it (snow forts, anyone?). If it sank, our relationship with ice would be vastly different, probably much more limited, and certainly less fun.
So, the next time you’re enjoying a cold drink and see those ice cubes floating, give them a little nod of appreciation. They’re not just keeping your beverage cool; they’re a testament to the remarkable, sometimes counterintuitive, properties of water that make our planet habitable. Pretty neat, huh? It's a little science lesson happening right there in your glass, and you didn't even have to take a test!

A Matter of Molecules and Mysteries
It’s fascinating to consider how a simple change in temperature can lead to such a profound change in the behavior of a substance. The transition from liquid to solid for water isn’t just a change of state; it’s a fundamental shift in its molecular arrangement and, consequently, its density.
This unique property of water isn't something you find in just any old liquid. Most substances become more compact and denser when they solidify. For example, iron gets denser when it freezes. If ice behaved like iron, our planet would be a very different place. Lakes and oceans would be frozen solid from the bottom up, and the entire basis of aquatic life would be nonexistent.
The hydrogen bonds in water are the unsung heroes here. They’re responsible for holding the molecules together, but in a way that creates space rather than packing them tightly. It’s this delicate balance of attraction and repulsion that gives water its peculiar, life-saving property.
The Everyday Miracle
It really is an everyday miracle, isn’t it? We pour ourselves a glass of water, toss in some ice, and we don’t even think about the physics at play. It's so commonplace that we rarely pause to consider the profound implications.
From the smallest organism surviving in a frozen pond to the global climate systems that regulate our weather, the fact that ice floats is a cornerstone of life on Earth. It's a subtle but incredibly powerful force of nature that shapes our world in ways we often take for granted.
So, the next time you’re reaching for that ice cube tray, remember this little anecdote. Remember the unique molecular dance that’s happening, the rebellion against the usual rules of physics, and the profound impact it has on our planet. It’s a tiny piece of science, served chilled, in every glass.