What Is The Electron Configuration For Ru3+

Alright, settle in, grab your latte, and let's talk about something that sounds way more intimidating than it is. We're diving into the electrifying world of electron configurations, specifically for our buddy, the Ruthenium ion with a charge of +3. Yeah, I know, sounds like something you’d find in a sci-fi movie script, but stick with me. Think of it as a chemistry gossip session, with a touch of existential dread about where all those tiny electrons are hiding. Because, let me tell you, these electrons are the ultimate ninjas of the universe, always on the move, and sometimes they just up and leave.
So, Ruthenium, what's the deal? Well, Ruthenium (let's just call him Ru for short, because saying "Ruthenium" three times fast is a workout) is a metal. A shiny, silvery metal. It's like the quiet, unassuming guy at the party who secretly has a killer dance move. It’s in that super-cool group of elements called the transition metals. These guys are the MVPs of the periodic table, playing crucial roles in everything from catalysts in industrial processes to being… well, let’s just say they’re involved in some important stuff.
Now, before Ru can become Ru3+, he’s just a regular, neutral atom of Ruthenium. And like any atom, he’s got a whole posse of electrons buzzing around his nucleus. These electrons aren't just randomly floating around; oh no. They have preferences. They like to hang out in specific "neighborhoods" called orbitals. Think of it like an apartment building with different floors and apartments. The floors are energy levels, and the apartments are the orbitals themselves: the 's' apartments (spherical, super chill), the 'p' apartments (dumbbell-shaped, a bit more dramatic), the 'd' apartments (way more complex shapes, like a fancy origami project), and even 'f' apartments (don't even ask, they're basically abstract art).
Must Read
The electron configuration tells us exactly how these electrons are distributed in these orbitals. It's like giving you the address of every single electron in the atom. For a neutral Ruthenium atom (atomic number 44, which means 44 protons and, you guessed it, 44 electrons), the full electron configuration is a bit of a mouthful: 1s2 2s2 2p6 3s2 3p6 4s2 3d10 4p6 5s1 4d7. Phew! That's a lot of electrons to keep track of. Imagine trying to organize a rave with 44 guests, each with their own favorite corner of the dance floor. It’s organized chaos, people!
But here’s where the drama kicks in: Ru3+. That little '+3' isn't just for show. It means our friend Ruthenium has gotten rid of three of his electrons. Electrons, remember, are negatively charged. So, when an atom loses negative charges, it becomes positively charged. It’s like losing your phone – you’re definitely less connected, and a bit more… bare.
![Ruthenium Electron Configuration: [Kr] 4d⁷ 5s¹ Explained](https://valenceelectrons.com/wp-content/uploads/2021/12/Electron-configuration-via-Aufbaw-principal.jpg)
Now, here's the really interesting part, and where things get a little tricky, like trying to explain quantum mechanics to your cat. When transition metals lose electrons to form ions, they don't lose them from the outermost shell first. Nope. They’re rebels. They lose them from the highest principal energy level first. This is because, even though the 'd' orbitals might be filled later in the configuration, their energy levels can sometimes be higher than the 's' orbitals in the next principal energy level when it comes to losing electrons. It’s like a hidden exit that’s easier to get to than the main door.
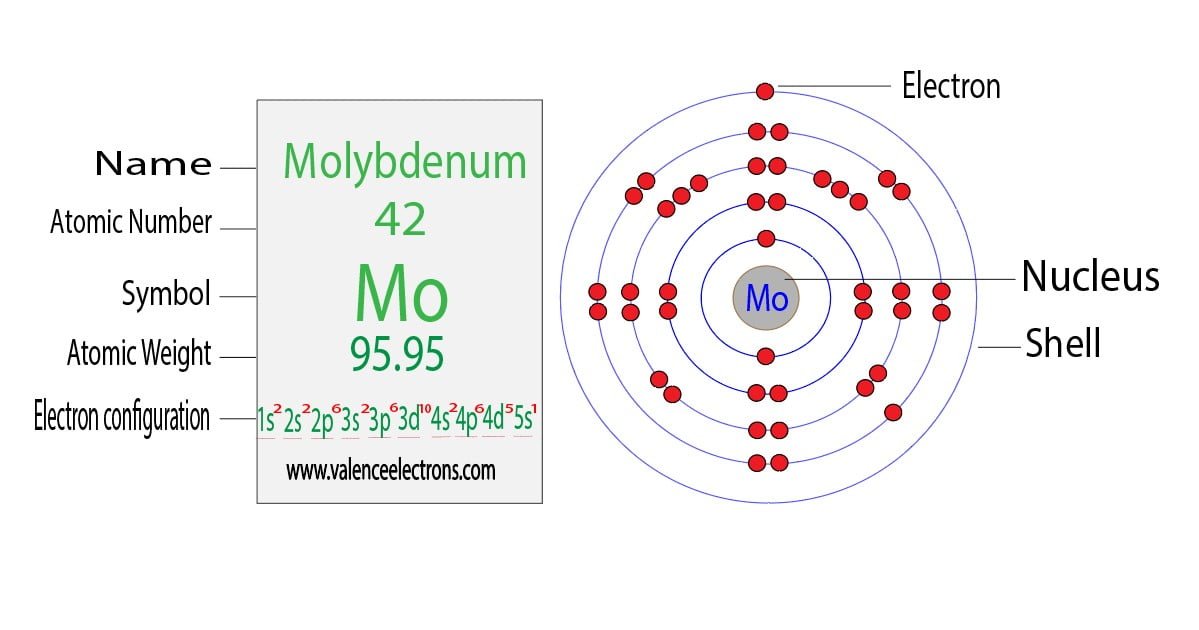
So, for our Ruthenium (Ru), the neutral configuration ends with 5s1 4d7. The highest principal energy level here is n=5, where the 5s orbital is. But the next highest is n=4, with the 4d orbitals. When Ruthenium becomes Ru3+, it first loses the electron from the 5s orbital. That’s one down, two to go. That makes it 5s0, or as we chemists sometimes say, "poof, it's gone."
![Ruthenium Electron Configuration: [Kr] 4d⁷ 5s¹ Explained](https://valenceelectrons.com/wp-content/uploads/2022/01/Ruthenium-Ru-orbital-diagram-768x404.jpg)
Then, it needs to lose two more. Where do they come from? They come from the 4d orbitals. So, the 7 electrons in the 4d orbitals become 5. So, our beloved 4d7 becomes 4d5.
And there you have it! The electron configuration for Ru3+ is: 1s2 2s2 2p6 3s2 3p6 4s2 3d10 4p6 4d5. Ta-da! It’s like a magic trick, but with less rabbits and more subatomic particles. We’ve basically stripped away the 5s electron and two of the 4d electrons. The 3d orbitals, which were completely full in the neutral atom (3d10), are left untouched, happily chilling like they’re in a spa day. They’re the real troopers!
Why is this important, you ask? Well, this electron configuration tells us a lot about how Ru3+ will behave. The number of electrons in the outermost shells, and particularly the unpaired electrons (if any), dictate its magnetic properties, its reactivity, and how it interacts with other atoms. For Ru3+, the 4d5 configuration means it has five unpaired electrons in its d orbitals. That’s a lot of unpaired electrons, which makes it paramagnetic. This means it’s attracted to magnetic fields, like a tiny, metallic magnet wannabe. Imagine tiny little magnets all lined up, trying to high-five each other when you bring a bigger magnet near. Cute, right?
So, next time you hear about Ruthenium or its ions, don't just glaze over. Remember our little electron shedding story. It's not just about memorizing numbers and letters; it's about understanding the quirky personalities of these invisible particles that make up everything around us. And who knows, maybe the next time you're struggling to find your keys, you can blame it on a rogue electron that decided to change its configuration. It's scientifically plausible… ish.

![Ruthenium Electron Configuration: [Kr] 4d⁷ 5s¹ Explained](https://valenceelectrons.com/wp-content/uploads/2023/03/Electron-Configuration-Calculator.jpg)