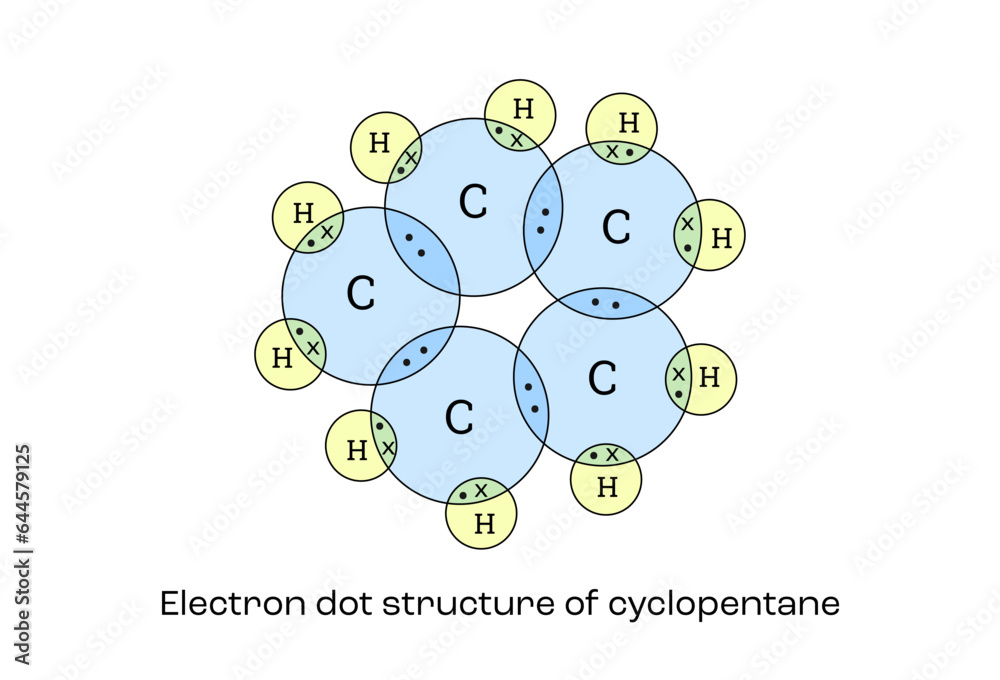
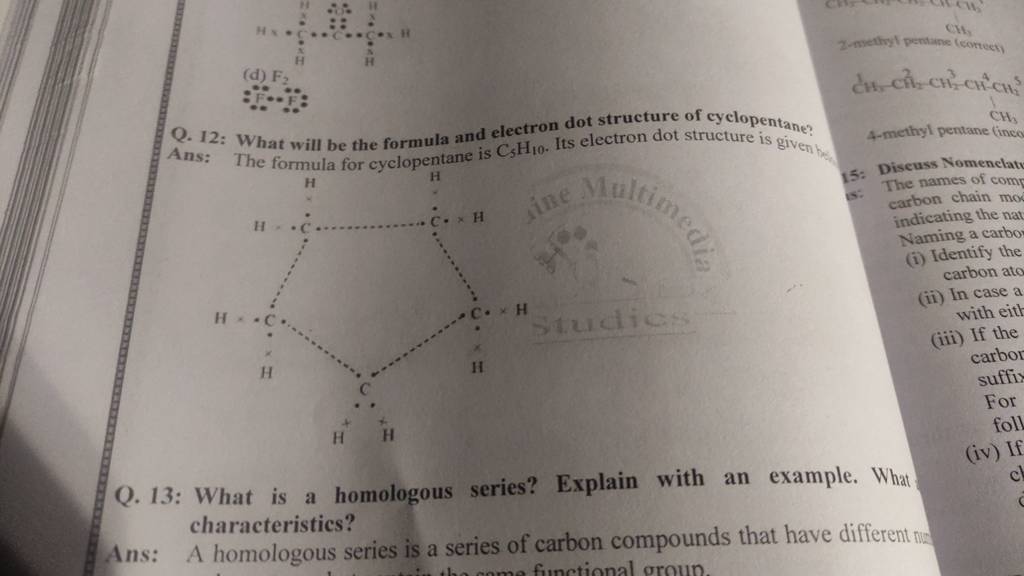
What Is A Proper Structure For Cyclopentane
Ever wonder what makes some molecules do their cool chemical tricks? It’s all about their shape! And when it comes to ring-shaped molecules, one of the most common and surprisingly interesting ones is cyclopentane. Think of it as the little black dress of organic chemistry – simple, elegant, and surprisingly versatile. But beneath its unassuming appearance lies a fascinating world of structural gymnastics that chemists find endlessly intriguing. So, why should you care about the structure of a five-carbon ring? Because understanding this seemingly simple molecule unlocks a deeper appreciation for how the world around us is built, from the fuels that power our cars to the complex molecules that make up our bodies.
The Perfect Five-Membered Ring: Not Quite Flat!
When you first imagine a molecule like cyclopentane, you might picture a perfectly flat, five-sided shape, like a pentagon drawn on a piece of paper. And in many simplified diagrams, that's exactly how it's shown! It's a handy shorthand. However, in the real, three-dimensional world of atoms and bonds, things are a little more dynamic and, dare I say, fun. A truly flat cyclopentane ring would be under a lot of strain. Imagine trying to hold a hula hoop perfectly flat with five people holding onto it at equal distances – it would want to buckle and twist! The carbon atoms in cyclopentane feel a similar pressure. This is due to something called angle strain. In a perfect flat pentagon, the angles between the carbon-carbon bonds would be 108 degrees. However, the ideal angle for carbon atoms bonded to four other atoms (like in cyclopentane) is closer to 109.5 degrees, as found in a tetrahedral arrangement. Trying to squeeze those bonds into 108-degree angles causes a bit of discomfort for the molecule.
To relieve this uncomfortable strain, cyclopentane doesn't stay flat. Instead, it adopts a shape that's a compromise, a clever way to get closer to that ideal tetrahedral angle without breaking apart. The most common and stable conformation, or shape, for cyclopentane is known as the envelope conformation. Imagine a five-pointed envelope, where one flap is bent upwards, and the other four lie more or less in a plane. This "bent" shape allows some of the carbon atoms to move slightly away from the planar arrangement, reducing the angle strain. It's like a dancer adjusting their pose to maintain balance and grace.
Must Read
The Envelope Conforms: A Slight Twist of Fate
In the envelope conformation, four of the carbon atoms are almost in the same plane, while one carbon atom is puckered out of this plane. This puckering is key. It’s not a drastic bend, but a subtle shift that makes a big difference. Think of it as a slight ripple on the surface of a still pond. The hydrogen atoms attached to the carbon atoms also play a role in this dance. They will be oriented either pointing "up" or "down" relative to the average plane of the ring. These positions are referred to as axial and equatorial, though in the dynamic envelope and its close relative, the half-chair conformation, these labels are less rigid than in larger rings like cyclohexane.
The envelope conformation isn't the only trick up cyclopentane's sleeve. It can also adopt a half-chair conformation. This is like a slightly more twisted version of the envelope, where two adjacent carbon atoms are puckered in opposite directions. Imagine a gently rocking chair – that’s a good visual for the half-chair. Both the envelope and half-chair conformations are constantly interconverting, meaning the molecule is rapidly switching between these slightly different shapes. This process is called ring flipping, and it happens very quickly at room temperature. It’s like a miniature molecular dance, with the ring bending and twisting to find its most comfortable position.
Why Does This Matter? The Practical Power of Shape
So, why go through all this trouble describing molecular wiggles? Because the shape of a molecule dictates how it interacts with other molecules. This is fundamental to chemistry and biology. For cyclopentane, its flexible ring structure and its ability to adopt different conformations affect its reactivity. For example, the way substituents (other atoms or groups attached to the ring) are oriented can influence how easily they can react or how they fit into the active site of an enzyme.

Think about it like trying to fit different-shaped puzzle pieces together. If a molecule is rigid and flat, it might only fit in one specific way. But if it's a bit flexible and can twist and bend, it has a better chance of fitting into various nooks and crannies, or reacting with different partners. This flexibility is incredibly important in the design of new drugs and materials. Scientists often study the conformations of molecules like cyclopentane to understand their behavior and to predict how they will behave in different chemical environments. It's also a building block for many more complex organic molecules, including some of the key components of our DNA and the fats that make up our cell membranes. So, the next time you see a simple five-membered ring, remember that it's not just a flat drawing, but a dynamic, shape-shifting marvel of molecular engineering!
The subtle dance of cyclopentane’s structure is a beautiful example of how even the simplest organic molecules are far from static. Their ability to bend and twist is crucial for their function.