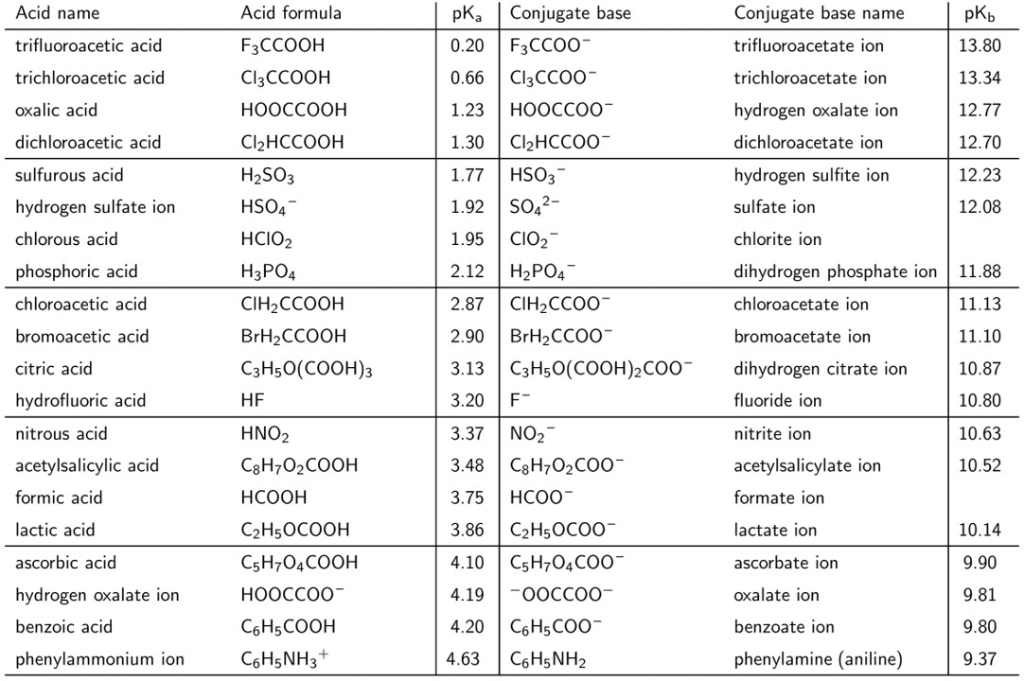
The Formula For The Conjugate Acid Of Hso3-

Alright, settle in, grab your imaginary latte, and let's dive into the wonderfully weird world of chemistry. Today, we're tackling a beast, a real head-scratcher, something that sounds like it belongs in a superhero origin story: the formula for the conjugate acid of HSO₃⁻. Don't let the jumble of letters and numbers scare you. Think of me as your friendly neighborhood chemistry guru, armed with bad jokes and a surprisingly good grasp of acid-base shenanigans.
So, what in the name of all that is slightly acidic is HSO₃⁻? Well, imagine a molecule that's been through a bit of a rough patch. It's got a sulfur atom doing its best to hold things together, surrounded by a few oxygen atoms, and then, the pièce de résistance, a hydrogen atom that's kind of on the fence. And that little negative sign on top? That means it's not feeling entirely neutral. It's like that friend who’s always a little bit dramatic, you know? They’ve lost a hydrogen ion, and they’re feeling the void.
Now, in the grand arena of chemistry, where everything is either an acid or a base (or, you know, something else that's just trying to get by), this HSO₃⁻ guy is what we call an amphoteric substance. This is a fancy word that basically means it can play on both teams. It can act like a base and accept a proton (which is just a fancy term for a hydrogen ion, H⁺), or it can act like an acid and give away a proton. It’s the ultimate chemical chameleon, folks! Like that one person at a party who can talk about quantum physics and then, five minutes later, is regaling you with tales of their cat’s latest shenanigans. Impressive, and a little confusing.
Must Read
But here’s the twist in our little chemical drama. We're not interested in HSO₃⁻ playing the acid card today. Oh no. We're here to find its conjugate acid. This is where things get interesting, and frankly, a little bit like detective work. Think of it as reverse engineering for molecules. We want to know who made HSO₃⁻ the way it is, or, more accurately, who could have donated that missing proton.
So, what's the golden rule of conjugate acids? It's simpler than you think, and I promise it doesn't involve a secret handshake or a cryptic riddle. To find the conjugate acid of any base, you simply have to do one thing: add a proton. That’s it! Just tack on an H⁺. It’s like giving it a little chemical hug, a warm welcome back into the proton-sharing club.
Let’s apply this super-secret, highly classified, top-tier chemical technique to our HSO₃⁻. We’ve got our dramatic HSO₃⁻, and we need to add an H⁺. What happens when you combine two things that both have hydrogen in them? You get more hydrogen, obviously! It’s like adding more sprinkles to an already sprinkle-covered cupcake – you just end up with more deliciousness.
The Grand Unveiling: H₂SO₃
So, if you take HSO₃⁻ and you add an H⁺, what do you get? Drumroll, please… H₂SO₃! Ta-da! We’ve found our conjugate acid. It’s called sulfurous acid, and it’s the slightly less dramatic, slightly more complete version of HSO₃⁻. Think of HSO₃⁻ as the sad sack who lost their keys, and H₂SO₃ as the happy camper who just found them again. Much better, right?

Let’s break down this transformation. Our original friend, HSO₃⁻, was feeling a little bit short on hydrogen. It had one hydrogen atom and a negative charge. When it encounters something that's happy to give up a proton (like water, which is a surprisingly generous soul in these situations, or even a stronger acid), it readily snatches that H⁺ right up. Poof! It becomes H₂SO₃. The negative charge disappears because the added positive charge of the proton perfectly cancels it out. It’s like a cosmic balancing act, a chemical détente.
This whole conjugate acid-base thing is super important in chemistry. It helps us understand how reactions proceed, how buffers work (those amazing substances that resist changes in pH, kind of like a chemical bodyguard), and why your stomach acid (which is mostly HCl, a very strong acid!) doesn’t eat you from the inside out. It’s all about these proton-swapping relationships.

Now, some of you might be thinking, "Okay, but what is sulfurous acid, H₂SO₃?" Well, it’s a real thing, though it's a bit of a weakling compared to its more famous cousin, sulfuric acid (H₂SO₄). You know, the one that sounds like it could dissolve a dragon. Sulfurous acid is often found in… wait for it… sulfur dioxide pollution. Yeah, that’s the stuff that makes smog and makes your lungs feel like they’ve been doing a thousand push-ups. So, while H₂SO₃ is the conjugate acid of HSO₃⁻, it’s not exactly the life of the party in an environmental sense. It’s more of a… cautionary tale.
But! Let’s not be too harsh. Sulfurous acid also plays a role in some food preservation and as a bleaching agent. It’s a bit of a Jekyll and Hyde molecule, capable of both mischief and helpfulness. Much like that one friend who accidentally sets off the fire alarm while trying to make toast, but then also manages to fix your computer.

So, there you have it. The formula for the conjugate acid of HSO₃⁻ is H₂SO₃. It’s a simple addition problem, really. Just remember: conjugate acid = base + proton. Easy peasy, lemon squeezy. Or, in this case, maybe more like sulfur-y, acid-y. See what I did there? I’ll be here all week. Try the veal.
And next time you’re feeling a bit short on protons, just remember the humble HSO₃⁻. It’s out there, waiting for a friendly H⁺ to complete its chemical destiny. It’s a testament to the fact that sometimes, all you need to become something greater is a little bit of help from a friend. Or, in this case, a proton.
The world of chemistry is full of these fascinating transformations, these subtle shifts in molecular identity. And at its heart, a lot of it comes down to who’s willing to give up a proton and who’s willing to take it. It’s the ultimate act of chemical generosity, or perhaps, chemical neediness. Either way, it makes for some seriously interesting science. So go forth, and conjugate with confidence!