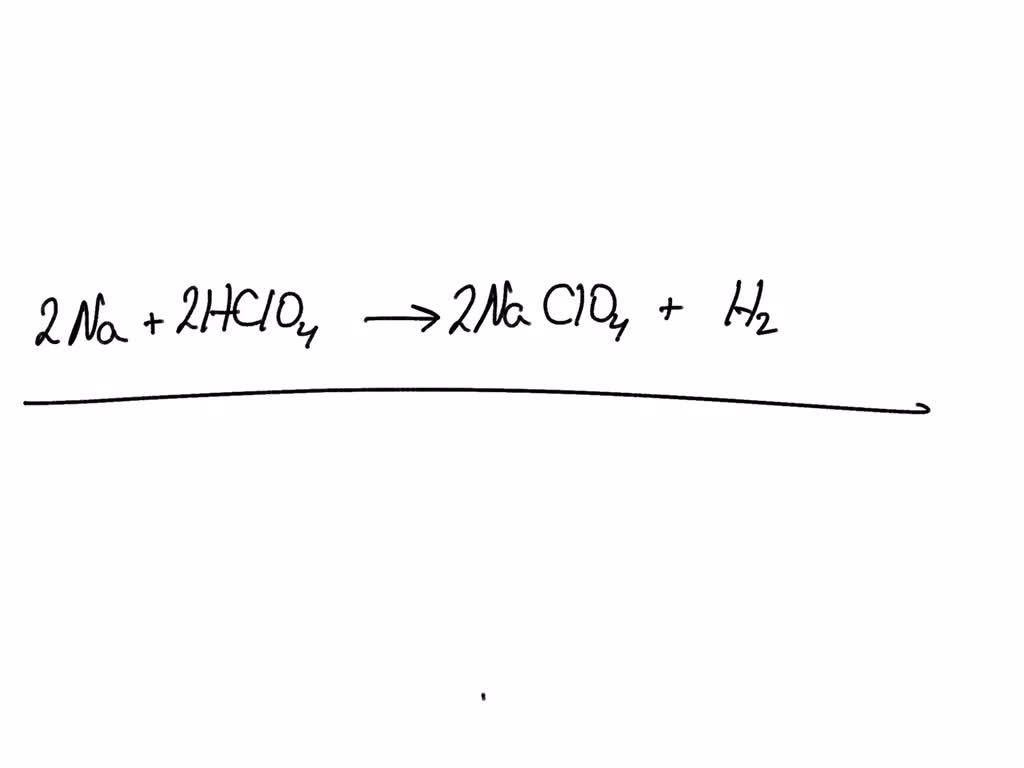Perchloric Acid And Water Balanced Equation

Alright, so let's chat about something that sounds way more intimidating than it actually is: perchloric acid and water. Yeah, I know, "perchloric acid" sounds like something a mad scientist would brew up in a spooky basement, maybe something that fizzes violently and turns your eyebrows green. And while it can be a bit feisty if you're not careful, when it hangs out with good old H₂O, things get a whole lot more chill. Think of it like that super intense friend who, when they finally relax and grab a coffee with you, turns out to be surprisingly down-to-earth.
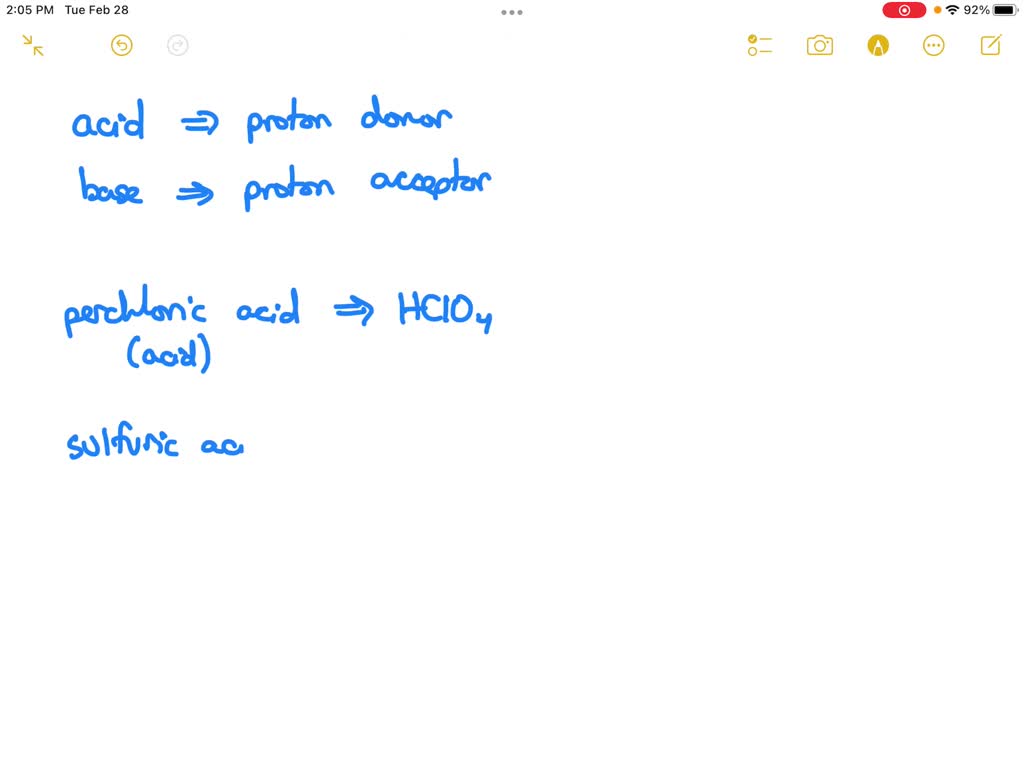
You see, in the grand ol' circus of chemistry, acids are basically the show-offs. They love to donate a proton, which is like a tiny, positively charged particle. It's their party trick, their whole raison d'être. And perchloric acid, HClO₄, is like the headline act. It's a really strong acid, meaning it's super eager to shed that proton. It's not shy about it, not one bit.
Now, water. Bless its simple, dihydrogen monoxide heart. Water is usually the supportive friend, the one who’s always there to soak things up and make everything less dramatic. It's like the comfy couch at the end of a long day, the fluffy towel after a splash in the pool. Water is really good at accepting protons. It's its superpower, in a way.
Must Read
So, when perchloric acid decides it's time to do its proton-donating thing, who's the first one to volunteer to catch it? You guessed it, water! It’s like when you’re handing out cookies at a party, and everyone’s reaching out. Perchloric acid is the one with the whole batch, and water is the eager guest with the biggest plate.
This is where the "balanced equation" part comes in. In chemistry, an equation is just a way of showing what happens when things react. It's like a recipe, but instead of flour and sugar, we're talking about molecules and atoms. A balanced equation means that for every atom or molecule you start with on one side, you end up with the exact same number of atoms or molecules on the other side. Nothing magically appears or disappears. It’s like saying if you start with two eggs and a cup of milk, you should end up with the ingredients for something that, when cooked, is still made of two eggs and a cup of milk, just in a different form. No disappearing eggs, right?
So, the balanced equation for perchloric acid (HClO₄) reacting with water (H₂O) looks like this:
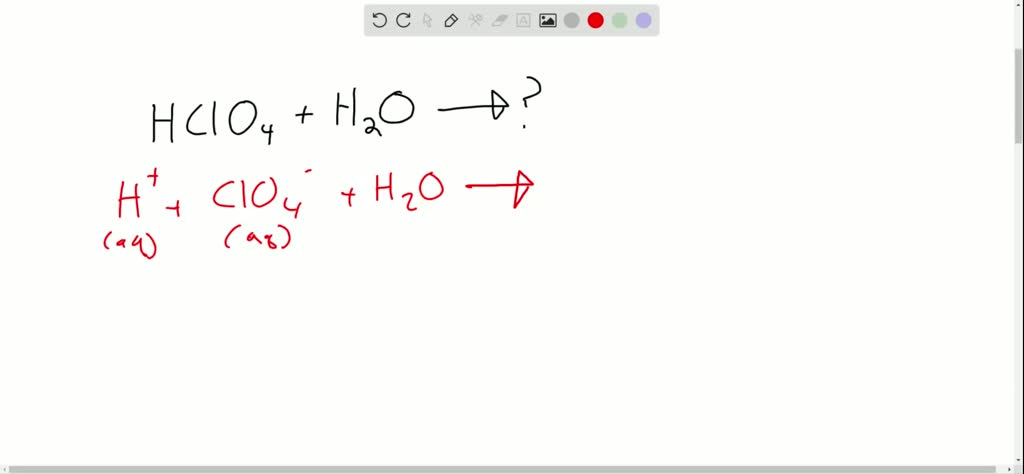
HClO₄ (aq) + H₂O (l) → H₃O⁺ (aq) + ClO₄⁻ (aq)
Whoa, hold on there, science whiz! Before your eyes glaze over, let's break that down like we're explaining it to your uncle who still thinks TikTok is a type of clock.
First off, the HClO₄ (aq). That's our headline act, perchloric acid. The (aq) just means it's dissolved in water, or "aqueous." Think of it like dissolving sugar in your tea. The sugar is still there, just spread out and making your tea taste sweet. So, the perchloric acid is happily mingling with the water molecules.
Then we have the + H₂O (l). That's our supportive friend, water. The (l) means it's in its liquid form, which is, you know, how we usually find water. It's just chilling, ready for action.

Now, the arrow (→). This is the "goes to" or "reacts to form" part. It’s the transformation, the plot twist. What happens after they meet?
On the other side, we have H₃O⁺ (aq). This is the really cool part. Remember how perchloric acid is all about donating a proton? Well, water is all about accepting it. When water (H₂O) grabs that extra proton (H⁺) from the perchloric acid, it becomes a hydronium ion. It's like H₂O got a little H buddy and became H₃O. It’s now a bit more crowded, a bit more energized. This is the acidic part of the solution. It's the reason why perchloric acid in water is, well, acidic.
And finally, we have the + ClO₄⁻ (aq). This is what’s left of the perchloric acid after it’s handed over its proton. It’s the perchlorate ion. It’s like the perchloric acid saying, "Alright, I gave away my best shot, but I'm still here!" The little minus sign (⁻) means it now has a negative charge. It lost a positive proton, so it's a bit unbalanced in the other direction. But don't worry, in the grand scheme of things, everything's still accounted for.
So, the whole equation is basically saying: "Perchloric acid, when mixed with water, politely gives a proton to a water molecule, forming a hydronium ion (which makes things acidic) and a perchlorate ion." It’s a calm exchange, a gentle handshake, not a wrestling match.
Why is this whole proton-sharing thing important? Well, it’s the fundamental process for how acids work in water. It’s why vinegar makes salad dressing tangy, and why that fizzy soda makes your tongue tingle. The H₃O⁺ ions are the culprits behind that characteristic sourness and reactivity of acids.
Think about making lemonade. You squeeze lemons (which contain citric acid), and then you add water and sugar. The citric acid molecules in the lemon juice, like our perchloric acid, are strong enough to interact with the water. They donate protons, and voila! You get that delicious, tart taste that’s actually a bunch of hydronium ions doing their thing.
Now, perchloric acid is a bit of a heavier hitter than citric acid. It’s like the difference between a friendly nudge and a firm pat on the back. It’s used in some pretty cool, and sometimes serious, applications. For instance, it's a key ingredient in rocket propellants. Yep, the stuff that sends rockets into space. It’s like the ultimate energy drink for those metal giants. And it’s also used in some etching processes in laboratories and in the production of certain explosives. So, while its reaction with water is simple, the resulting solution can be quite powerful.
But even with its powerful applications, the initial act of dissolving in water is incredibly common. It’s like how a superhero has their secret identity, or how a powerful engine has its starting mechanism. The balanced equation is that gentle start, the way things begin before they get to their more dramatic performances.
It’s important to remember that while the reaction itself is balanced, perchloric acid still needs to be handled with respect. It’s not something you’d want to spill on your favorite shirt, unless you want a permanently bleached spot the size of a frisbee. It's a strong oxidizer, meaning it loves to steal electrons from other things, which can lead to some exciting, and potentially hazardous, reactions if it comes into contact with organic materials. Think of it as having a very enthusiastic roommate who tends to tidy up by throwing everything in the bin, whether you want it thrown or not.
The beauty of chemistry, though, is that even the most potent substances have fundamental rules governing their behavior. And the reaction between perchloric acid and water is a classic example of acid-base chemistry in action. It's a dance between a giver and a receiver, a transfer of a tiny particle that has big consequences for how things taste, how things react, and how things get done.
So, next time you hear about perchloric acid, don't immediately picture a bubbling cauldron. Picture a strong acid, a willing water molecule, and a simple, balanced exchange. It's a foundational moment, the first step in a chemical journey that can lead to anything from a delicious tartness to the awe-inspiring power of a rocket launch. It's a reminder that even the most complex scientific processes often start with the simplest, most elegant interactions. It’s like a handshake that sets the stage for an epic adventure.
And that, my friends, is the wonderfully chill, surprisingly relatable story of perchloric acid and its balanced equation with water. Just a little bit of proton sharing, a lot of chemical harmony, and a whole lot less scary than you might have thought. Now, if you’ll excuse me, I think I need a glass of water. It’s amazing how much it does for us, isn’t it?