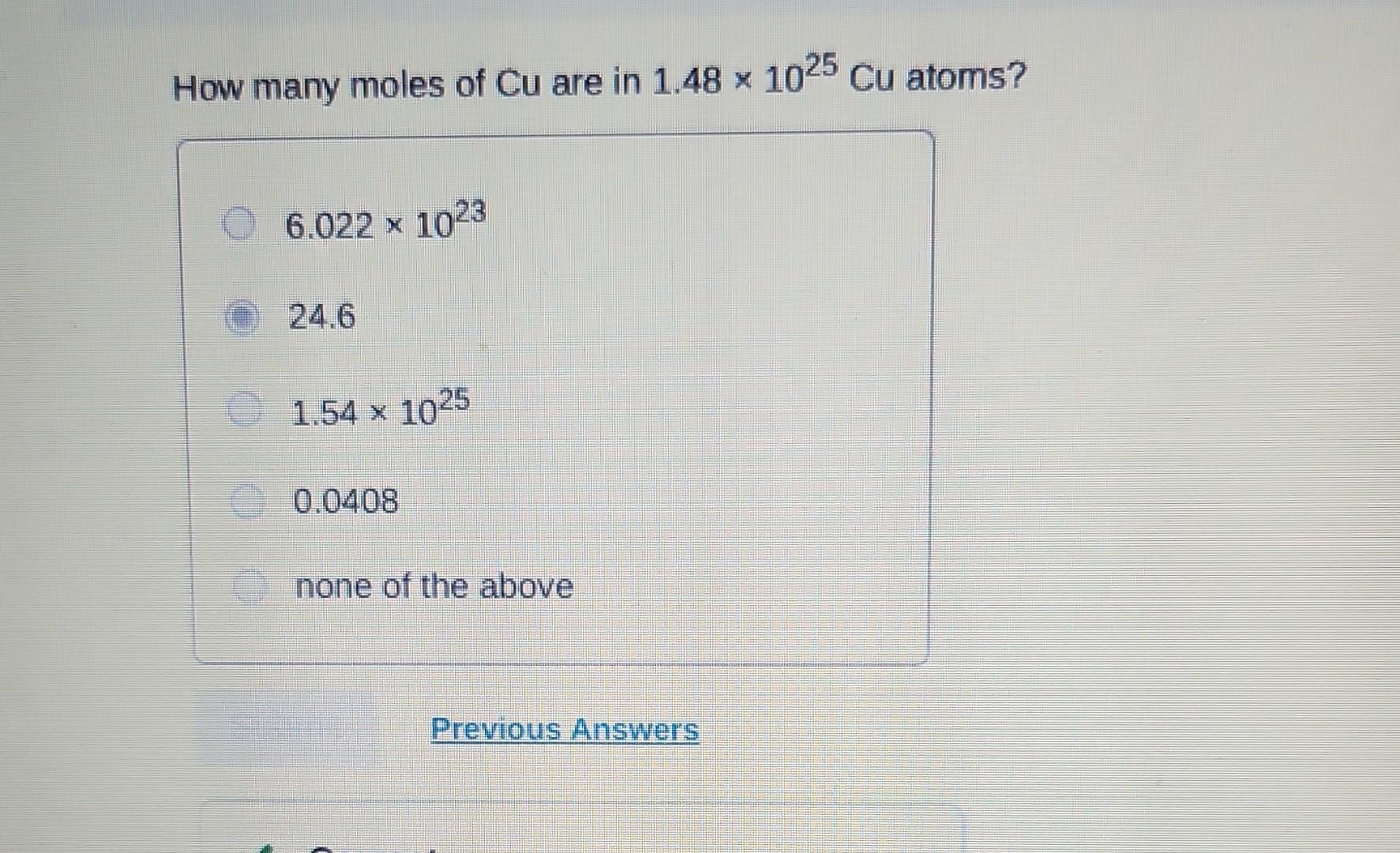
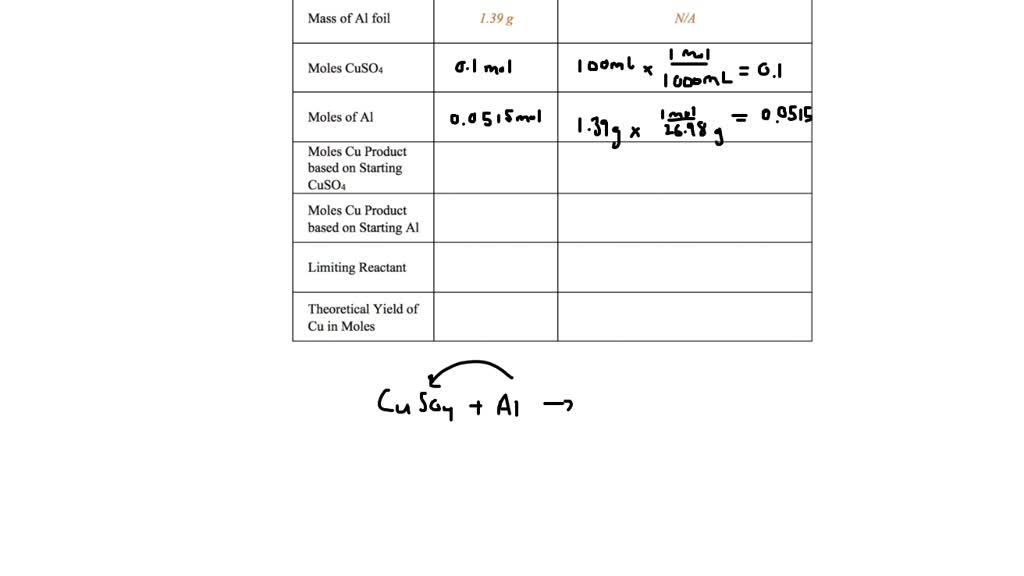
Moles Of Cu Product Based On Starting Cuso4
Hey there, curious cats and science dabblers! Ever found yourself staring at a bag of something that looks a bit like blue sand and wondered, "What's this all about?" Well, buckle up, because we're about to dive into a world that sounds a bit intimidating – "Moles of Cu Product Based on Starting CuSO4" – but is actually as down-to-earth as figuring out how many cookies you can bake from a bag of flour. Yep, we're talking about something pretty neat, and it all starts with that delightful blue stuff, copper sulfate, or CuSO4 as the science folks like to call it.
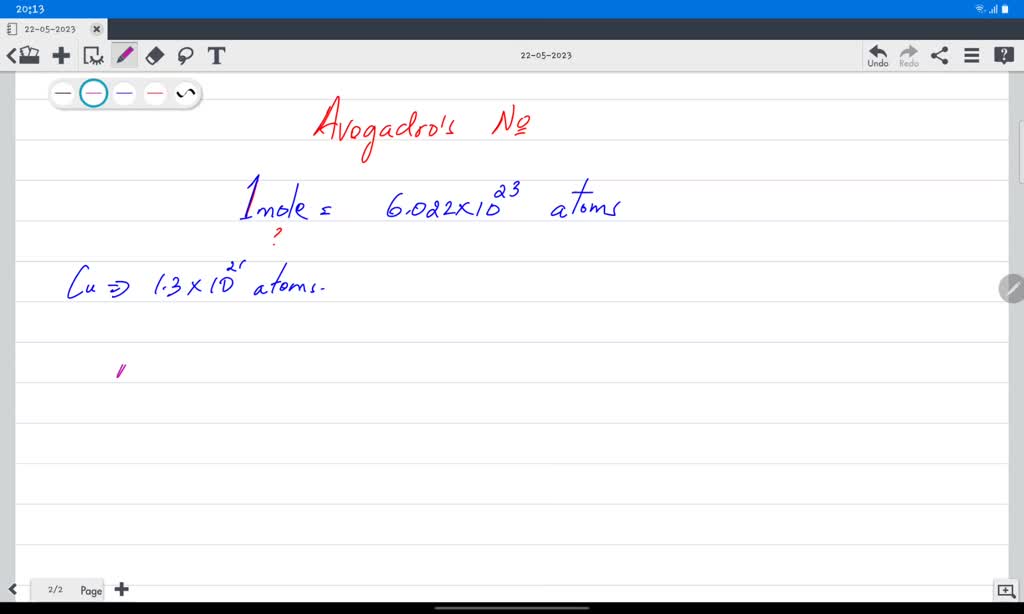
Think of it like this: you've got a recipe for cookies, right? The recipe tells you how much flour you need, how many eggs, and how much sugar. If you want to make, say, 24 cookies, you know you need a certain amount of each ingredient. If you suddenly decide you want to make 48 cookies (who wouldn't?), you double all your ingredients. Simple as that. Well, in the world of chemistry, this "recipe" idea is super important. And when we talk about "moles," we're basically talking about a really big number that chemists use to count tiny things like atoms and molecules. It's like having a giant scoop that holds exactly 6.022 x 1023 of something. That's a 1 with 23 zeros after it! Enough to make your head spin, but trust me, it's a handy way to keep track.
So, when we say "moles of Cu product based on starting CuSO4," we're essentially asking: "If I start with a certain amount of our blue copper sulfate (CuSO4), how much copper (Cu) can I make?" Copper, as you probably know, is that reddish-brown metal that makes your pennies (well, the old ones!), your electrical wires, and even some really cool decorative items. It's a pretty useful element, and knowing how much of it we can get from our starting blue stuff is like knowing how many delicious cookies you'll end up with from your bag of flour. It's all about the yield, the outcome, the what-you-get-out-of-it part of the chemical equation.
Must Read
Why Should You Care About This Blue Sand and Copper?
Okay, I hear you. "Why should I, a regular person who probably worries more about what's for dinner than molecular counts, care about this?" Great question! It turns out, this seemingly complex concept is at the heart of a lot of things we encounter every single day. Think about it:
Your Phone's Inner Workings: That smartphone in your pocket? It's packed with copper wires. The electricity that powers it, that lets you scroll through endless cat videos, travels through copper. Knowing how to efficiently make copper from its compounds like CuSO4 is crucial for producing all those tiny components.

The Pretty Blue Stuff Around Us: Ever seen those vibrant blue crystals in science kits or as decorative items? That's often copper sulfate! But it's also used in other ways. For example, it can be used to kill algae in ponds or as a fungicide in agriculture. Understanding how much copper is in that blue stuff, and what else you can get from it, is important for its safe and effective use.
The Art of Making Things: From jewelry to statues, copper has been used by artists for millennia. Whether it's a new artist experimenting with creating their own patinas or a large-scale industrial process, knowing the precise amount of copper you can extract from a source is vital for controlling the quality and cost of the final product.
Environmentally Friendly Solutions: Sometimes, chemical processes can be a bit… messy. By understanding the "recipe" of how to get copper from CuSO4, scientists can figure out the most efficient and least wasteful ways to do it. This is like figuring out how to bake your cookies without burning half the batch or leaving a mountain of dirty dishes. It's about being smart and sustainable.

Let's Get a Little More Specific (But Still Chill!)
Imagine you're a baker with a special recipe. Your recipe calls for 100 grams of flour to make, say, 10 perfect muffins. Now, let's say your "flour" is actually our blue copper sulfate (CuSO4), and your "muffins" are pure copper (Cu). The chemical reaction is like the recipe. It tells you exactly how much flour (CuSO4) you need to get a specific number of muffins (Cu).
In chemistry-speak, the "mole" is our unit of measurement for these ingredients. If you have 1 mole of CuSO4, it contains a specific number of CuSO4 units. And through a chemical transformation, that 1 mole of CuSO4 can be converted into a certain amount of copper (Cu). The "certain amount" is usually expressed in moles too. So, the question "moles of Cu product based on starting CuSO4" is really asking: "If I start with X moles of CuSO4, how many moles of Cu will I end up with?"
This ratio, this conversion factor, is determined by the chemical reaction itself. It's like the recipe telling you that for every 100 grams of flour, you get 10 muffins. In chemistry, it's usually a very precise ratio. For instance, in a common process, 1 mole of CuSO4 can theoretically produce 1 mole of Cu. So, if you start with 2 moles of CuSO4, you could potentially get 2 moles of Cu. It’s a one-to-one swap in this simplified scenario!
A Little Story: The Case of the Lost Copper Coins
Let's say you have a little treasure chest filled with 5 moles of our lovely blue copper sulfate. You're on a mission to extract as much pure copper as possible to make some shiny new coins. You follow your trusty chemical recipe. If the recipe dictates that every mole of CuSO4 gives you exactly 1 mole of Cu, then from your 5 moles of CuSO4, you'd expect to get… 5 moles of Cu! It's like having 5 bags of flour, and each bag is guaranteed to give you a certain number of muffins. No surprises, just consistent results if you follow the process correctly.
But what if your recipe is a bit different? Maybe it's a more complex dessert, and for every 2 moles of CuSO4 you use, you only get 1 mole of chocolate chips (our copper!). In that case, from your 5 moles of CuSO4, you'd get a little less copper – around 2.5 moles. This is where understanding the exact chemical reaction and its ratios becomes super important. It’s the difference between perfectly portioned ingredients and a cake that might be a bit too dense or a bit too airy.

Why This Precision Matters (Even if You're Not a Chemist)
This "recipe" knowledge is crucial for industries. Imagine a company that makes copper wiring. They buy large quantities of copper compounds, like CuSO4. They need to know exactly how much copper they can get from each batch of starting material. If they underestimate, they won't have enough copper to meet demand, and customers will be unhappy. If they overestimate, they might be wasting money on raw materials that don't translate into actual copper.
It’s like a craft brewery. They know that for every pound of hops they use, they get a certain flavor profile and a specific amount of beer. They have to be precise to make sure every batch of their popular IPA tastes exactly the same. If their calculations were off, some batches might be too bitter, and some might not have enough punch. Consistency is key, and it all starts with understanding the ingredients and the process.
So, the next time you see those beautiful blue crystals or think about the wires in your electronics, remember that behind it all, there's a fascinating world of chemical recipes. And understanding "moles of Cu product based on starting CuSO4" is just one of the many ways scientists and engineers make sure we have the materials we need, efficiently and effectively. It’s a little bit of everyday magic, powered by the precise counting of tiny things!

.png)