How Many Valence Electrons Does Barium Have

Okay, so imagine you're at a super fancy party. Like, really fancy. And everyone's showing off their bling. Well, in the world of atoms, that bling is called valence electrons. They're the super important outer electrons. The ones that get to mingle and make chemical bonds. Think of them as the party animals of the atomic world!
Now, our star for today is an element called Barium. Sounds a bit like a fancy dessert, right? Barium tart! But nope, it's an element. And it's a pretty cool one, if you ask me. We're going to dive into its valence electrons. Because why not? It's way more exciting than doing laundry, I promise.
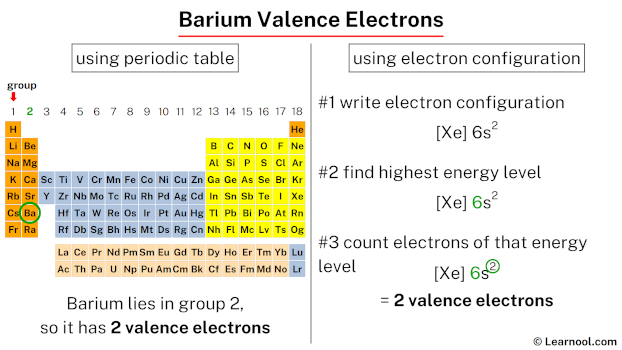
So, How Many Valence Electrons Does Barium Have?
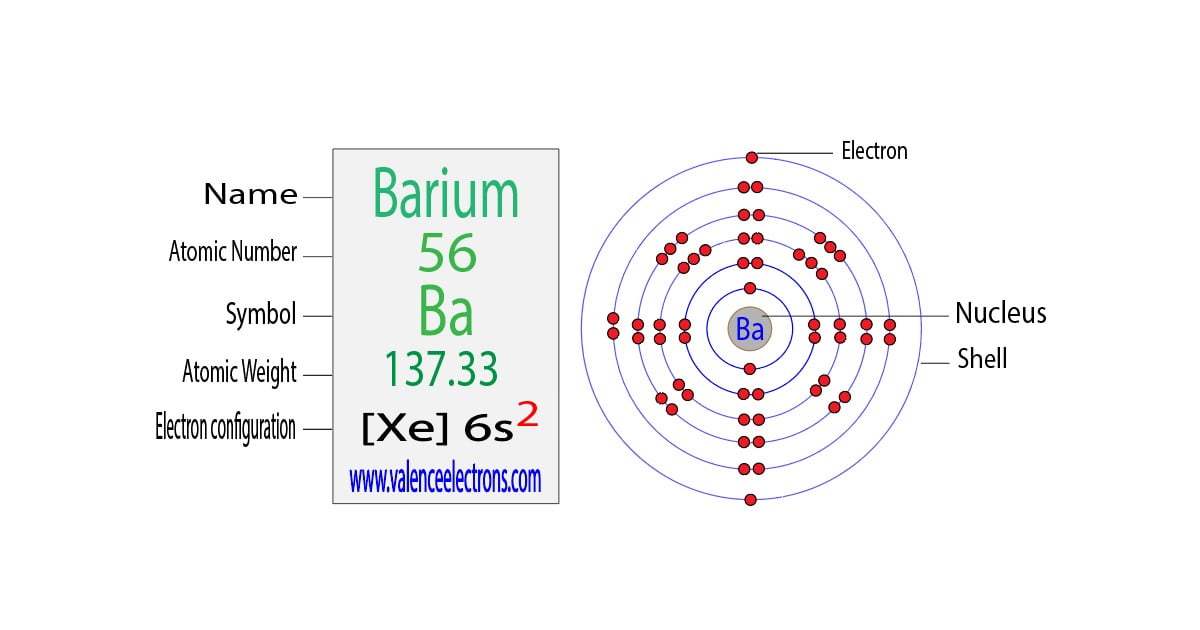
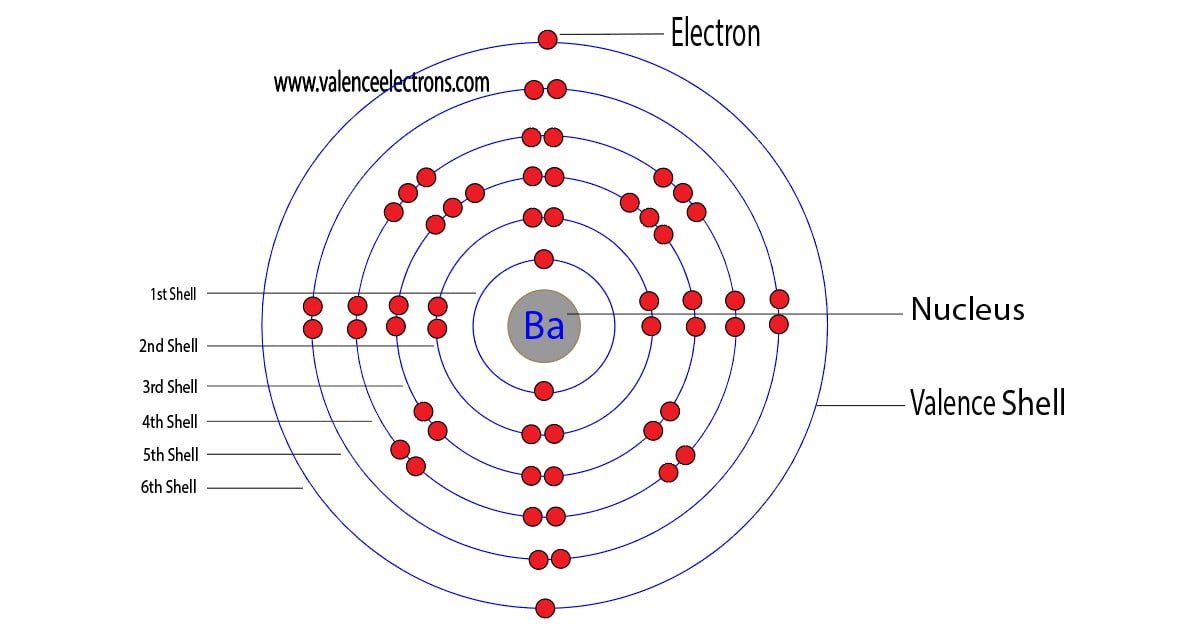
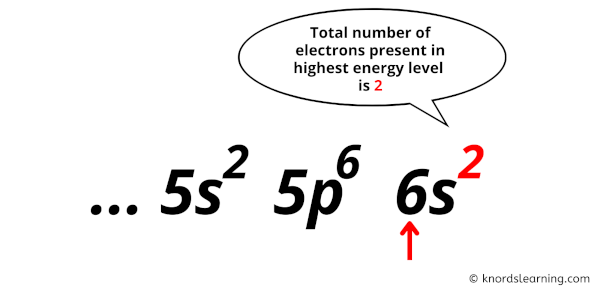
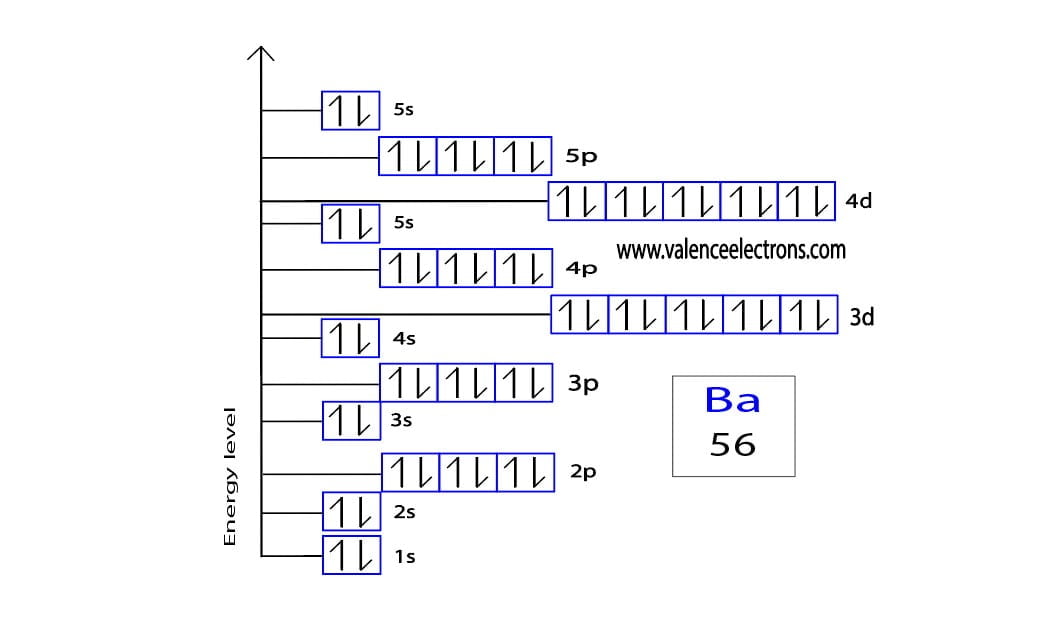
Get ready for it. The big reveal. Barium has… two valence electrons!
Must Read
Just two! That’s it. That’s the magic number. And honestly, that’s why Barium is so interesting. It’s like the element equivalent of someone who shows up to a party with just two perfect dance moves. But those two moves? They’re good. They’re effective.
Why only two? Well, it all has to do with where Barium hangs out on the periodic table. It’s in Group 2. And for most elements in Group 2, those two outer electrons are their signature. Their whole vibe.
The Periodic Table: It's Not Just a Bunch of Squares!
You might think the periodic table is just this boring chart in chemistry class. Wrong! It’s actually a giant family tree for elements. And the rows and columns? They tell you a ton of stuff. Barium’s position is like its address. And its address tells us its family secrets. Like how many valence electrons it’s got.

Barium is an alkaline earth metal. Fancy name, right? Alkaline earth. Sounds like a place you’d find some mystical creatures. But it just means it’s a type of metal. And these guys are known for being pretty reactive. Thanks to those two easily-given-away valence electrons.
Think of it this way: those two outer electrons are like loose change. Barium is just itching to get rid of them. It wants to be stable, like a noble gas with a full outer shell. So, it’s always looking for a buddy to take those electrons off its hands.
Why Are Valence Electrons Such a Big Deal?
This is where it gets really fun. Valence electrons are the architects of the chemical world. They’re why things stick together. They’re why you can have water, or salt, or that amazing chocolate cake you had last week.

When Barium meets up with another element, those two little valence electrons are the first ones to get involved. They’re the handshake. The first greeting. They decide if a bond will happen, and what kind of bond it will be.
Because Barium only has two, it’s a bit of a generous soul. It’s usually happy to give both of them away to an element that really wants them. This makes Barium form what we call ionic bonds. It’s like Barium is saying, “Here, take them! I don’t need them. Just don’t come back asking for more.”
Barium: The Drama Queen of Reactivity?
Now, Barium isn’t just giving away electrons to anyone. It’s a bit of a discerning character. It likes to react with things that are greedy for electrons, like nonmetals. Especially those in Group 17, the halogens. Those guys are practically begging for electrons!
When Barium meets a halogen, like chlorine (think of chlorine in swimming pools, but way more intense in element form!), it’s a match made in chemical heaven. Barium hands over its two electrons, and the halogen grabs them. Poof! A new compound is formed. It’s a chemical love story!
One of Barium’s most famous compounds is barium sulfate. You might have heard of it. It’s used in medical imaging, like X-rays. The barium makes your insides show up on the scan. Pretty cool, huh? All thanks to those two little valence electrons.
Quirky Barium Facts to Make You Smile
Let’s sprinkle in some fun stuff. Did you know Barium compounds can actually make things glow in the dark? Well, not exactly glow in the dark like your old Halloween decorations, but some Barium compounds are used in fireworks to create a brilliant green color! Imagine Barium at a party – it’s not just giving away electrons, it’s putting on a light show!
And here’s a slightly spooky one: pure Barium is actually quite dangerous. It’s highly reactive and can be toxic. So, while those two valence electrons are super important for chemistry, you probably don’t want to go around touching pure Barium. Stick to the chemistry books and maybe a nice barium tart for dessert.
But even with its dangerous side, Barium’s tendency to lose those two electrons is what makes it so useful. It’s a classic example of how an element’s electron configuration dictates its behavior. It’s like having a superpower, and Barium’s superpower is being a super generous electron donor.
So, What’s the Takeaway?
The next time you hear about Barium, you can impress your friends by saying, “Oh yeah, Barium! It’s got two valence electrons!” And then you can explain, in your most enthusiastic voice, how those two electrons are the key to its whole personality. They make it reactive, they let it bond with other elements, and they’re responsible for cool things like fireworks and medical scans.
It’s a simple number, two. But in the world of atoms, those two valence electrons make Barium a true star. It’s a reminder that even the smallest details in science can lead to big, fascinating outcomes. So, let’s give a cheer for Barium and its awesome two valence electrons! They’re the unsung heroes of the chemical universe. And honestly, talking about them is way more fun than dusting.