How Many Protons And Electrons Does S2 Have

Hey there, curious minds! Ever found yourself staring at a cloudy sky, a twinkling star, or maybe even just a perfectly ripe piece of fruit, and wondered about the tiny, invisible building blocks that make it all happen? Well, today, we're going on a little adventure into the surprisingly fun world of… atoms! And specifically, we're going to tackle a question that might sound a bit intimidating at first glance: how many protons and electrons does S2 have?
Now, before your eyes glaze over and you start thinking about complicated chemistry equations, let's take a deep breath and remember that learning is an adventure, not a chore! Think of it like trying to solve a fun puzzle. And this particular puzzle piece, S2, is actually quite fascinating. So, grab your metaphorical magnifying glass, and let's dive in!
What's This "S2" Thing Anyway?
So, what exactly is S2? In the grand, glorious theatre of chemistry, S is the symbol for an element called sulfur. You know, that stuff that can smell a bit… pungent sometimes? (Think rotten eggs – not exactly a perfume, is it? But super important for life!) Now, when you see that little number "2" after the S, it means we're not just talking about one sulfur atom, but two sulfur atoms that have decided to buddy up and form a molecule. It's like two friends holding hands and saying, "Hey, let's do this together!"
Must Read
This little partnership, this S2 molecule, is actually what makes up a good chunk of the air we breathe. Pretty cool, right? It's floating around us all the time, being an essential part of our atmosphere. So, when we ask about the protons and electrons in S2, we're really asking about the sum total in those two collaborating sulfur atoms.
Protons: The Atom's Heartbeat
Let's start with protons. Think of protons as the tiny, positively charged heart of an atom. They live right there in the center, in the nucleus, along with neutrons (which are like the neutral, steady companions). The number of protons an atom has is its defining characteristic. It's like its unique fingerprint, telling us exactly which element it is. For instance, every single atom with 6 protons is carbon. Every atom with 8 protons is oxygen. You get the idea!
![[Solved]: How many electrons, protons and neutrons does Sr^(](https://media.cheggcdn.com/study/761/761bebe4-4226-4139-8197-27e3f2130144/4-24-2502.01.jpeg)
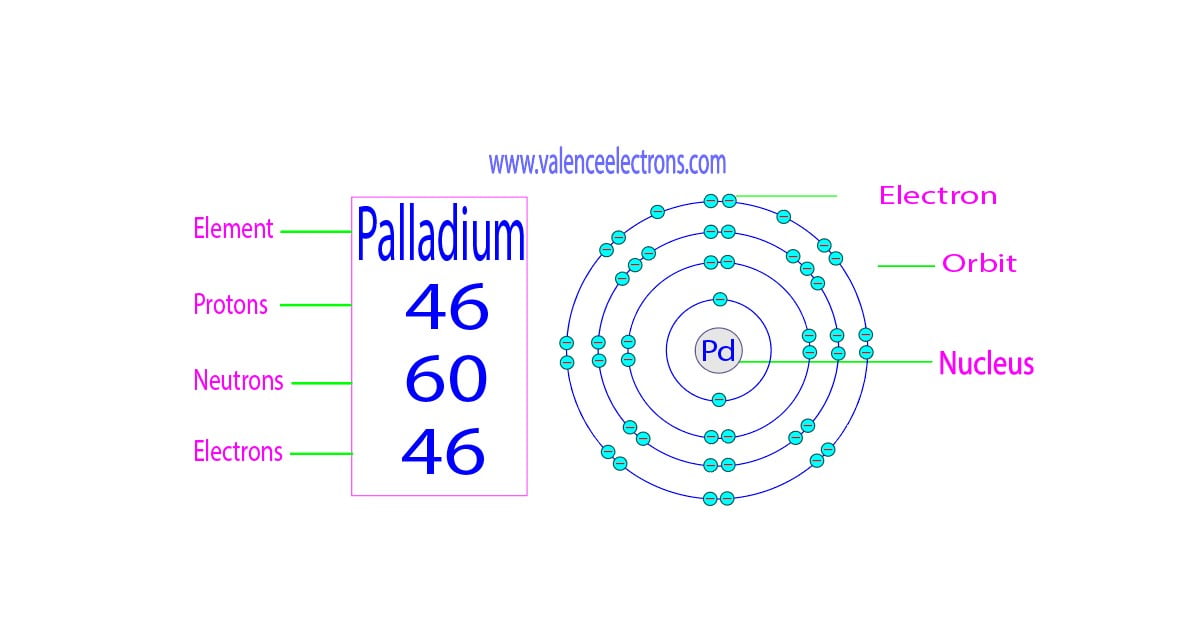
So, what about sulfur? Our friend, the element symbol S, has 16 protons. Yep, that's its atomic number, its identity card. And since S2 is made of two sulfur atoms, we have to do a little bit of simple math. If one sulfur atom has 16 protons, then two sulfur atoms will have 16 protons + 16 protons. That brings us to a grand total of 32 protons in our S2 molecule!
Isn't that neat? We've just uncovered a fundamental piece of information about something that's literally all around us! Thirty-two tiny, positively charged particles working together in those two sulfur atoms. It’s like a miniature universe of positive energy!
Electrons: The Speedy Dancers
Now, let's talk about electrons. These little guys are the negatively charged whirlwinds that zip around the nucleus. They're much smaller and lighter than protons, and they're constantly in motion. Think of them as the energetic dancers of the atom, moving in specific patterns and energy levels.

Here’s a super important rule in the atomic world: for a neutral atom (meaning it’s not carrying an extra electrical charge), the number of electrons is always equal to the number of protons. It’s like a perfect balancing act. Positive and negative charges cancel each other out, making the atom stable and content.
So, since each sulfur atom has 16 protons, a neutral sulfur atom will also have 16 electrons. And because our S2 molecule is made of two such sulfur atoms, we do our little math dance again! 16 electrons + 16 electrons equals a whopping 32 electrons in our S2 molecule!
Wowza! So, in that humble S2 molecule, we have a total of 32 protons and 32 electrons. They're all dancing around, holding hands, and creating the stuff that makes up our world. It’s a beautiful, intricate ballet of subatomic particles happening constantly, even as you read this!
Why Does This Even Matter? (Spoiler: It's Fun!)
You might be thinking, "Okay, that's a number. So what?" Ah, but that's where the fun truly begins! Understanding these numbers, these fundamental properties of elements and molecules, is like unlocking a secret language of the universe. It’s the key to understanding why things behave the way they do.
For example, the way these electrons are arranged in their orbits around the nucleus is what dictates how atoms interact with each other. It’s why sulfur can bond with oxygen to form sulfuric acid (a super important industrial chemical!), or why it plays a role in the taste and smell of certain foods. It's all down to the number and behavior of those tiny electrons!
Knowing about protons and electrons helps us appreciate the incredible complexity and beauty of even the simplest things. It’s the foundation for everything from medicine and materials science to understanding the stars and our own bodies. It’s a constant reminder that there’s so much more to the world than meets the eye.
The Joy of Discovery
So, the next time you encounter something that involves sulfur, maybe even just a whiff of that distinctive scent, you can smile and know a little secret. You know that S2 molecule is made up of a total of 32 protons and 32 electrons, all working in perfect harmony. It's a tiny piece of knowledge, perhaps, but it's a gateway to a much bigger understanding.
The world of science, from the smallest atom to the largest galaxy, is an endless source of wonder and discovery. And the best part? It's all accessible to you. You don't need a fancy lab coat or a PhD to start exploring. All you need is a spark of curiosity and a willingness to ask "why?" and "how?"
So, go ahead! Ask more questions. Look up other elements. Tinker with your understanding of the universe. Every new piece of knowledge you gain is like adding another vibrant color to your personal palette of understanding. Keep that curiosity alive, keep exploring, and never underestimate the incredible joy and inspiration that comes from simply learning something new!