How Many Moles Of Silver Atoms Are In 1.8

Hey there, science adventurers and curious minds! Ever looked at something shiny and wondered about the tiny, invisible building blocks that make it up? We’re talking about atoms, the superstars of the universe, and today, we’re diving headfirst into the dazzling world of silver! Get ready for a super fun exploration into how many of these little silver wonders are hiding in a seemingly innocent 1.8 grams of this precious metal.
Now, 1.8 grams might not sound like much. It’s less than a sugar cube, way less than your average smartphone, and barely enough to make a single, tiny silver charm. But oh, my friends, what it lacks in size, it makes up for in sheer, mind-boggling numbers of atoms!
The Tiny Titans of Silver
Imagine you have a giant jar, like, the biggest jar you can possibly envision. And in that jar, you're pouring in tiny, microscopic silver beads. We're not talking about the kind of beads you'd string for a bracelet, oh no. These are atoms, so small they make a speck of dust look like a boulder.
Must Read
To even begin to grasp the number we're talking about, we need a special kind of counting tool. Scientists, those brilliant brains, have invented a unit called the mole. Think of a mole as a super-duper dozen. A dozen eggs is 12. A mole of anything is... well, it’s a number so enormous it’ll make your head spin faster than a disco ball!
This special number, this "mole," is 6.022 x 1023. Let that sink in for a second. That's a 6 followed by 23 zeros. If you had a mole of grains of sand, they would stretch across the entire Earth multiple times, reaching way past the moon and back! It’s like counting all the stars in the universe and then counting them again, and then some more!
The Grand Unveiling: How Many Moles?
So, we have our 1.8 grams of sparkling silver. And we know that one mole of silver atoms weighs a specific amount. It’s like knowing that one box of a particular kind of toy weighs 5 pounds. If you have 10 pounds of those toys, you know you have two boxes.
Scientists have figured out that one mole of silver atoms weighs approximately 107.87 grams. This magical number is called the molar mass of silver. It’s like the weight of one of those giant mole-sized bags of silver atoms.
Now, we're not talking about 107.87 grams of silver. We’re talking about a much smaller, but still incredibly packed, 1.8 grams. We need to see how many of those "bags" (or rather, how many fractions of those bags) fit into our 1.8 grams.
It’s a bit like asking: if a giant pizza has 8 slices, and you only eat 2 slices, what fraction of the pizza did you eat? You ate 2/8, or 1/4 of the pizza. We’re doing something similar here, but with a much, much, much bigger denominator!

To find out how many moles are in 1.8 grams of silver, we simply divide the mass we have (1.8 grams) by the mass of one mole of silver (107.87 grams per mole). This is like saying, "How many times does the weight of one mole fit into our amount?"
And the answer, my friends, the wonderfully mind-boggling answer, is approximately...
0.0167 moles!
Yes, you read that right! It's not a whole number. It's a tiny fraction of a mole. But don't let that fool you. This seemingly small number represents an absolutely gargantuan amount of silver atoms!

The Atom Avalanche
Let's put this into perspective. If 1.8 grams of silver is a measly 0.0167 moles, what does that actually mean in terms of individual silver atoms? We take our fraction of a mole and multiply it by that HUGE number we talked about earlier: 6.022 x 1023 atoms per mole.
So, 0.0167 * (6.022 x 1023) = ... drumroll, please ...
Approximately 1.005 x 1022 silver atoms!
That’s a 1 followed by 22 zeros! If you were to write out that number, your paper would be a mile long! It's a number so big, it makes the national debt look like pocket change. It’s more numbers than there are people on Earth, more numbers than there are freckles on every single person who has ever lived. It's an atom-apalooza!
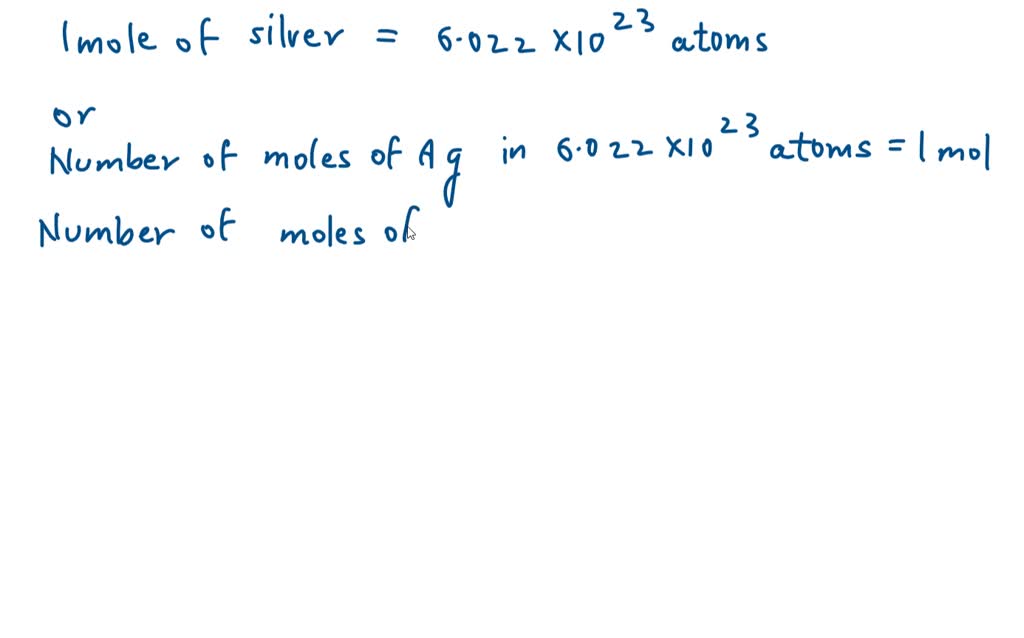
Think about it. Every single one of those 1.8 grams is packed, bursting at the seams, with these incredibly tiny, shimmering silver atoms. It’s like having a microscopic city, bustling with trillions upon trillions of little silver citizens, all doing their atomic thing, contributing to the shine and luster we admire.
So, the next time you see something made of silver, a beautiful ring, a vintage coin, or even a shiny spoon, take a moment to appreciate the sheer, unimaginable abundance of atoms contained within. That small piece of metal is a universe in itself, a testament to the incredible power and scale of the microscopic world.
It’s truly astonishing to think that such a small, everyday object can harbor such an astronomical number of fundamental particles. It makes you feel both incredibly small and strangely connected to the grand cosmic dance of matter. Science, my friends, is all about uncovering these hidden wonders, and today, we’ve uncovered a whole lot of silver!
So, go forth and marvel at the world around you! Even the smallest things hold the biggest secrets, and with a little bit of math and a whole lot of enthusiasm, we can start to unravel them, one atom, one mole, at a time. Isn't science just the coolest?





