How Are Neutral Atoms Converted Into Cations
Hey there, curious minds! Ever wonder what happens when things get a little… charged up? Today, we're going to dive into a super cool, yet surprisingly simple, concept in the world of atoms: how do neutral little guys turn into something called a cation? Think of it as atoms getting a tiny, energetic personality boost. Pretty neat, right?
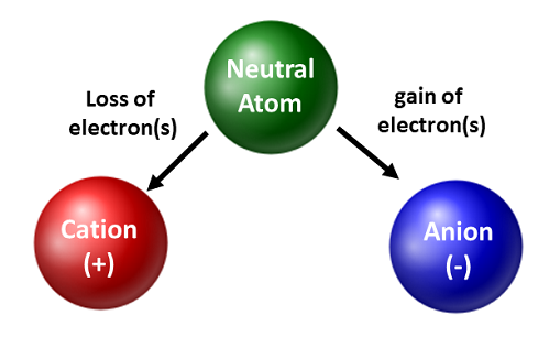
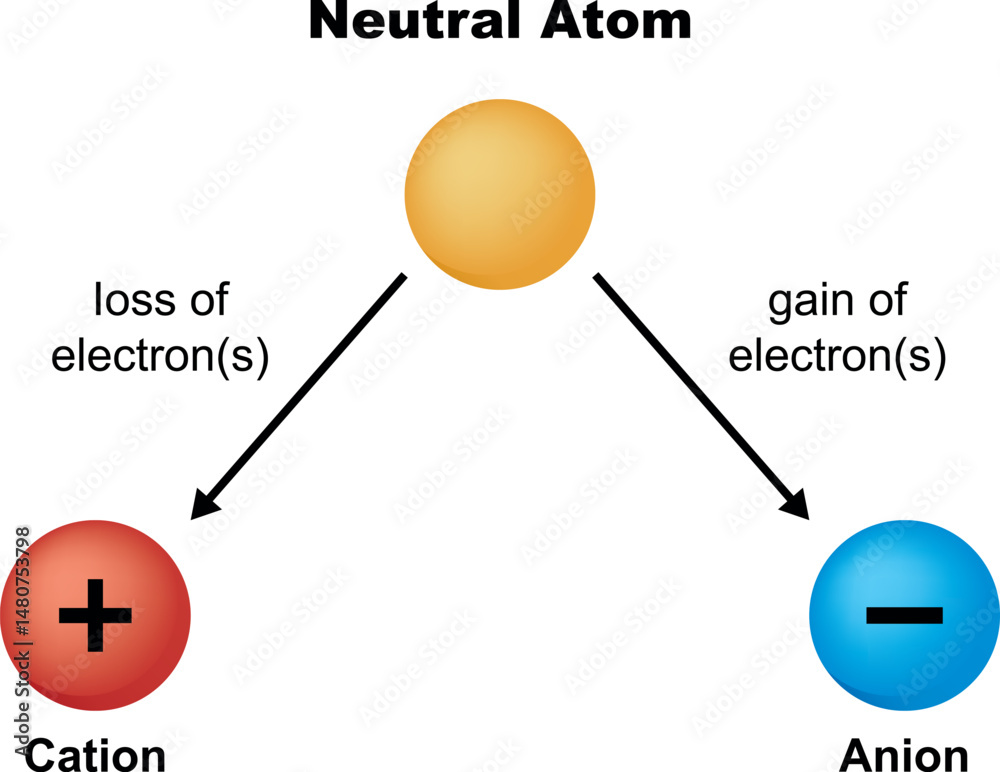
So, let's set the scene. Imagine an atom, just chilling. It's perfectly balanced, like a perfectly still pond. In this state, it's called neutral. Why neutral? Because it has an equal number of two key players: positively charged protons and negatively charged electrons. These guys are like the yin and yang of the atomic world, perfectly canceling each other out. No net charge, no drama.
But what happens when things aren't so perfectly balanced anymore? What if an atom decides to shed a little weight, or maybe pick up a new friend? That's where the magic of becoming a cation comes in. It's all about playing a little game of give and take with those pesky electrons.
Must Read
The Grand Exchange: How Electrons Go Rogue
You see, atoms aren't always stuck in their neutral state. Sometimes, they're feeling a bit unstable, like a juggling act that's about to drop a ball. This usually happens when they're interacting with other atoms. And what's the easiest part for an atom to let go of, or to grab onto? Yep, you guessed it: electrons.
Protons are pretty much locked away in the atom's core, the nucleus. They're not going anywhere easily. Electrons, on the other hand, are zipping around the outside, like little satellites in orbit. They're much more accessible, and a lot more willing to join a new party or leave an old one.

So, when an atom is in a situation where it needs to achieve a more stable electron arrangement (think of it as reaching a comfy, low-energy state, like finding the perfect spot on the couch), it might decide to lose one or more of its electrons. It's like saying, "You know what? I don't need all these extra little negative charges floating around. I'm going to be lighter and happier without them."
The "Loss" Leads to a Positive Vibe
Now, here's the mind-blowing part. If an atom starts with, say, 10 protons (which are positive) and 10 electrons (which are negative), it's perfectly neutral. Total charge? Zero.
But then, it decides to lose one electron. So now it has 10 protons and only 9 electrons. What happens to the balance? Suddenly, the positive charges have won! There's one more positive charge than negative charge. It's like having 10 positive points and only 9 negative points. The net result is a positive charge. And voilà! This atom is no longer neutral. It has become a cation.

Think of it like this: Imagine a tug-of-war. The protons are the strong anchors on one side, and the electrons are the team on the other. If the electron team gets smaller by one player, the proton team has a clear advantage. The whole rope (the atom) is now pulled in the positive direction.
This is why cations are always associated with a positive charge. They've actively given away something negative. It's a bit counter-intuitive, right? You might think losing something would make you feel weaker, but in the atomic world, sometimes shedding those negative electrons is what makes you stronger and more stable!

Why Do Atoms Even Bother? Stability, Baby!
So, why would an atom go through the trouble of losing electrons? It's all about finding that sweet spot of stability. Atoms, much like us, crave a sense of order and low energy. They look at their outer electron shells, and if they're not "full" in a certain way, they can be a bit… antsy.
Different atoms have different "preferred" numbers of electrons in their outermost shell. It's a bit like having a favorite number of guests at a party. If an atom has too many electrons in its outer shell for its liking, it's much happier to get rid of the excess. By losing these electrons, it can expose a more stable, "fuller" inner electron shell. It's like tidying up a messy room to feel more at peace.
This drive for stability is the fundamental reason behind so many chemical reactions. Atoms are constantly trying to achieve this happy, low-energy state. And one of the most common ways they do it is by becoming ions – either cations (by losing electrons) or anions (by gaining electrons, but that's a story for another day!).

You see these cations everywhere, even if you don't realize it. Think about the salt you put on your food. That's sodium chloride (NaCl). The sodium atom (Na) loses an electron to become a sodium cation (Na+), and the chlorine atom gains that electron to become a chloride anion (Cl-). These oppositely charged ions are then attracted to each other, forming the stable crystal structure of salt. It's like atomic matchmaking!
The "Cool" Factor of Cations
So, to recap, a neutral atom is like a perfectly balanced scale. It has an equal number of positive protons and negative electrons. When an atom loses one or more of these negative electrons, it ends up with more positive charges than negative ones. This imbalance gives it a net positive charge, and bam, it's now a cation!
It's a fundamental process that underpins so much of the chemistry we see around us. From the electricity flowing through wires to the biological processes happening inside your body, cations play a vital role. They're the charged-up little particles that make things happen. Pretty amazing for something so tiny, don't you think? Next time you see something happen in the world, remember that somewhere, an atom might have just decided to get a little positively charged!




![[Solved] When a neutral atom is converted in to cation, there is](https://storage.googleapis.com/tb-img/production/20/10/F1_Aman_Madhu_23.10.20_D3.png)