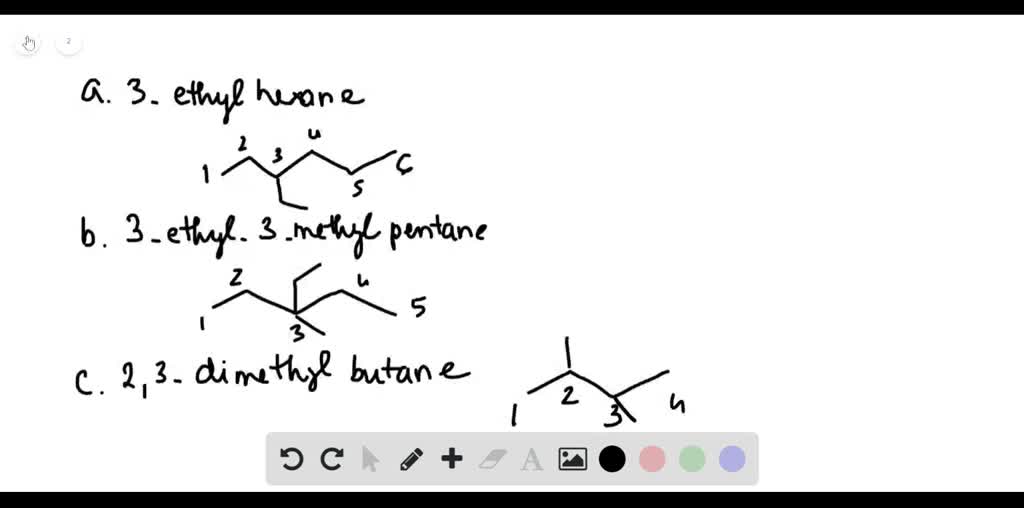
Draw A Structural Formula For 3-ethylhexane.

Alright, chemistry explorers, gather 'round! Today, we’re diving into the wonderfully wacky world of organic molecules, and I promise you, this is going to be easier and more fun than trying to fold a fitted sheet. We're going to tackle drawing the structural formula for a molecule called 3-ethylhexane. Think of it as giving our little molecule a stylish makeover, complete with all its bits and bobs perfectly placed. No need to panic; this is less like a pop quiz and more like building with LEGOs, but with atoms and bonds!
First things first, let’s break down that name, 3-ethylhexane. It’s like a secret code, and once you crack it, the whole drawing process becomes as clear as a freshly washed window. The “hexane” part is our foundation. Imagine it as a long, straight chain of six carbon atoms. Carbon atoms are the building blocks of organic chemistry, the ultimate party animals of the molecular world, always linking up with others. So, picture six of these carbon buddies holding hands in a row. That’s your basic hexane skeleton. Easy peasy, right? It’s like drawing a line of six dots. We’re going to represent these carbon atoms with the letter 'C', but sometimes, in structural formulas, we get a bit lazy (in a good, efficient way!) and just use the corners and ends of lines to show where they are. Think of it as the ‘implied presence’ of a carbon atom. Revolutionary!
Now, for the “ethyl” part. This is where things get a little more interesting, a little more flamboyant. An ethyl group is like a little side hustle for our main chain. It’s made up of two carbon atoms, and it’s going to be attached to our hexane chain. But where? Ah, that’s where the “3” comes in! This number tells us exactly where our ethyl group needs to make its grand entrance. It’s going to attach itself to the third carbon atom in our hexane chain.
Must Read
Think of the hexane chain as a row of houses numbered 1 through 6. The '3-ethyl' tells us that a little two-carbon house (the ethyl group) is connected to the third house in the row. It's like adding a fancy porch to your bungalow!
So, let’s visualize this. We’ve got our six carbons in a row. We number them mentally, one, two, three, four, five, six. At position number three, we’re going to stick on that ethyl group. Remember, the ethyl group is two carbons. So, from that third carbon on our main chain, we’re going to draw a little arm reaching out, and at the end of that arm, we’ll have another carbon atom. And that second carbon atom in the ethyl group? It needs a friend too, so it’ll be connected to the first carbon of the ethyl group. It’s like a mini-chain sprouting from the main highway!
Now, let’s talk about the invisible helpers: the hydrogen atoms. These little guys are the loyal companions of carbon atoms. Every carbon atom loves to be connected to a total of four other atoms (these can be other carbons or hydrogens). Our main hexane chain, with its six carbons, will have some hydrogens attached to satisfy these connections. For carbons at the end of the chain (carbons 1 and 6), they’ll be connected to one other carbon, so they’ll need three hydrogen atoms each. For the carbons in the middle (carbons 2, 4, and 5), they are connected to two other carbons, so they’ll need two hydrogen atoms each.
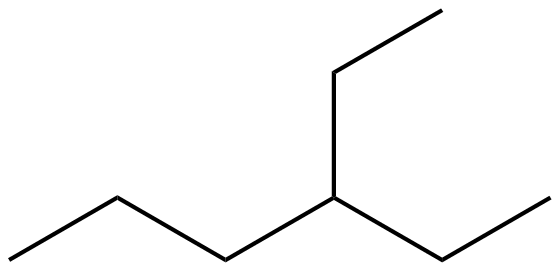
But what about carbon number 3? This is where our ethyl group is hanging out. Carbon 3 is connected to carbon 2, carbon 4, and the first carbon of our ethyl group. That’s three connections already! So, it only has room for one hydrogen atom. Pretty neat, huh? And our ethyl group? The first carbon in the ethyl group is connected to carbon 3 of the hexane chain and the second carbon of the ethyl group. So, it needs two hydrogen atoms. The second carbon of the ethyl group is only connected to the first carbon of the ethyl group, so it needs three hydrogen atoms.
When we draw structural formulas, we often simplify things. We might not draw every single hydrogen atom explicitly. Instead, we can use lines to represent the bonds between atoms. Each line is a bond. A single line is a single bond, and atoms that aren't explicitly shown are assumed to be carbon atoms. So, our hexane chain can be drawn as a zig-zag line with six points (or ends and corners). Then, at the third point from either end, we’ll draw another shorter zig-zag line (representing the two-carbon ethyl group) branching off. This skeletal structure shows all the carbon atoms and how they're connected. The hydrogen atoms are then understood to be there, filling in the remaining bonding spots for each carbon.
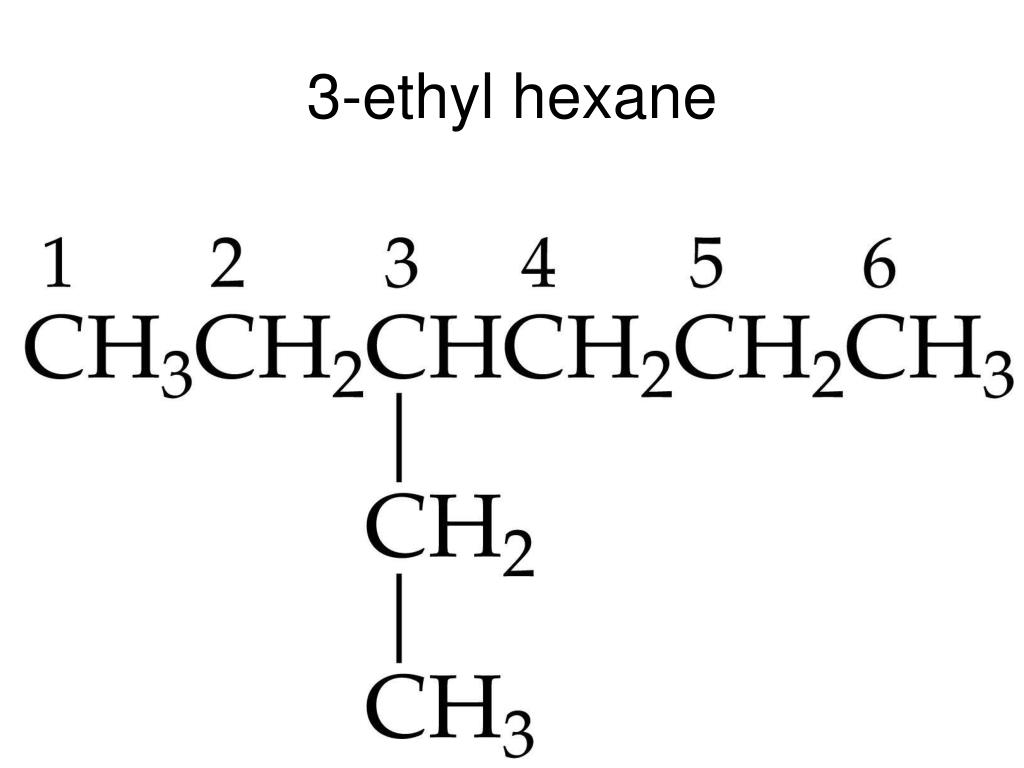
So, imagine that zig-zag line for hexane. You number it (mentally, or you can even lightly sketch numbers if you’re a beginner – no judgment here!). At number three, you draw another little zig-zag sticking out. That’s your 3-ethylhexane! It's a beautiful, albeit simple, piece of molecular architecture. It’s like a main road (hexane) with a little scenic route (ethyl group) branching off at a specific junction. And all those hydrogens are like the invisible passengers making sure everyone is comfortable and has enough room!
Drawing these formulas is like creating a map of the molecule. The longer the name, the more intricate the map, but the underlying principles are always the same: count your carbons, identify your longest chain, and then attach any extra bits according to the numbers. It’s a fantastic way to understand how molecules are built and how their structures influence their properties. So, next time you hear a chemical name, don’t shy away. Embrace it as a fun puzzle, and you'll be drawing structural formulas like a pro in no time. You’ve got this!