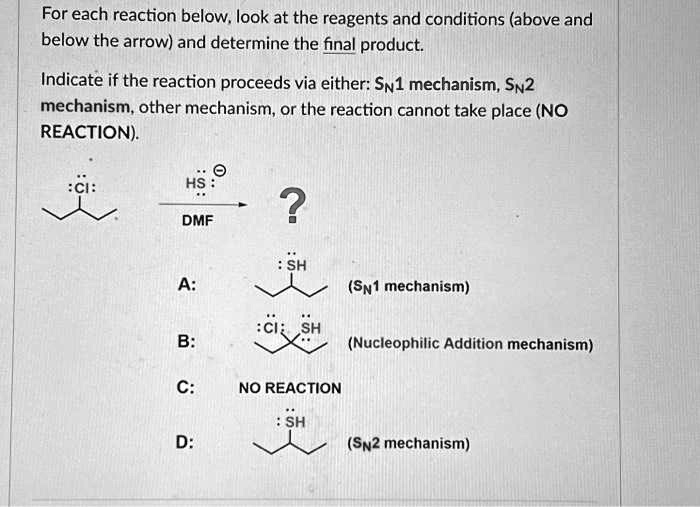
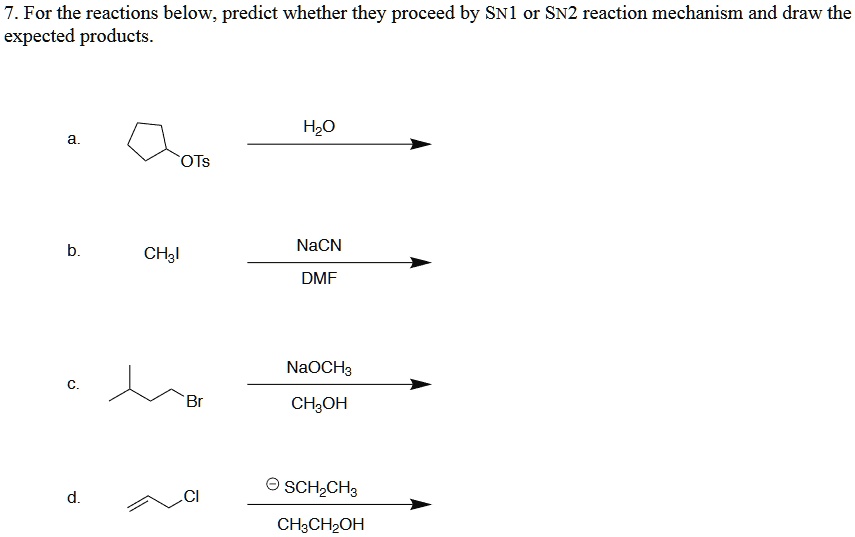
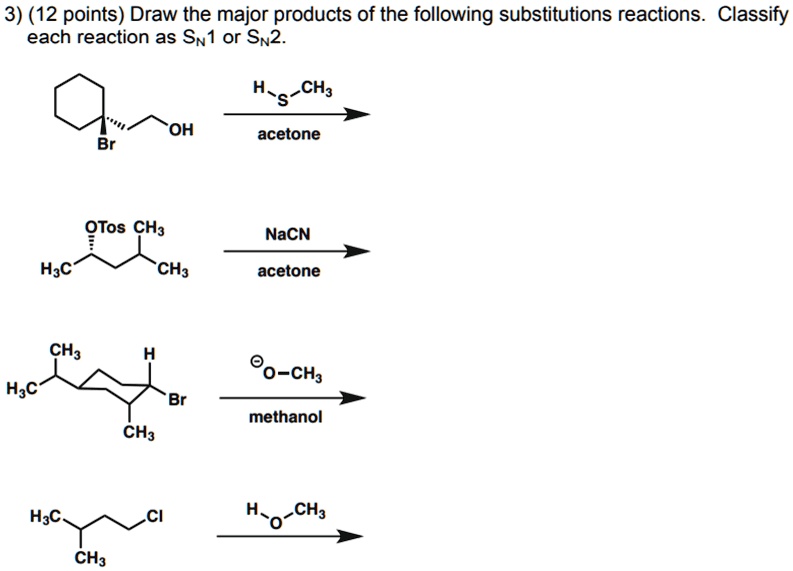
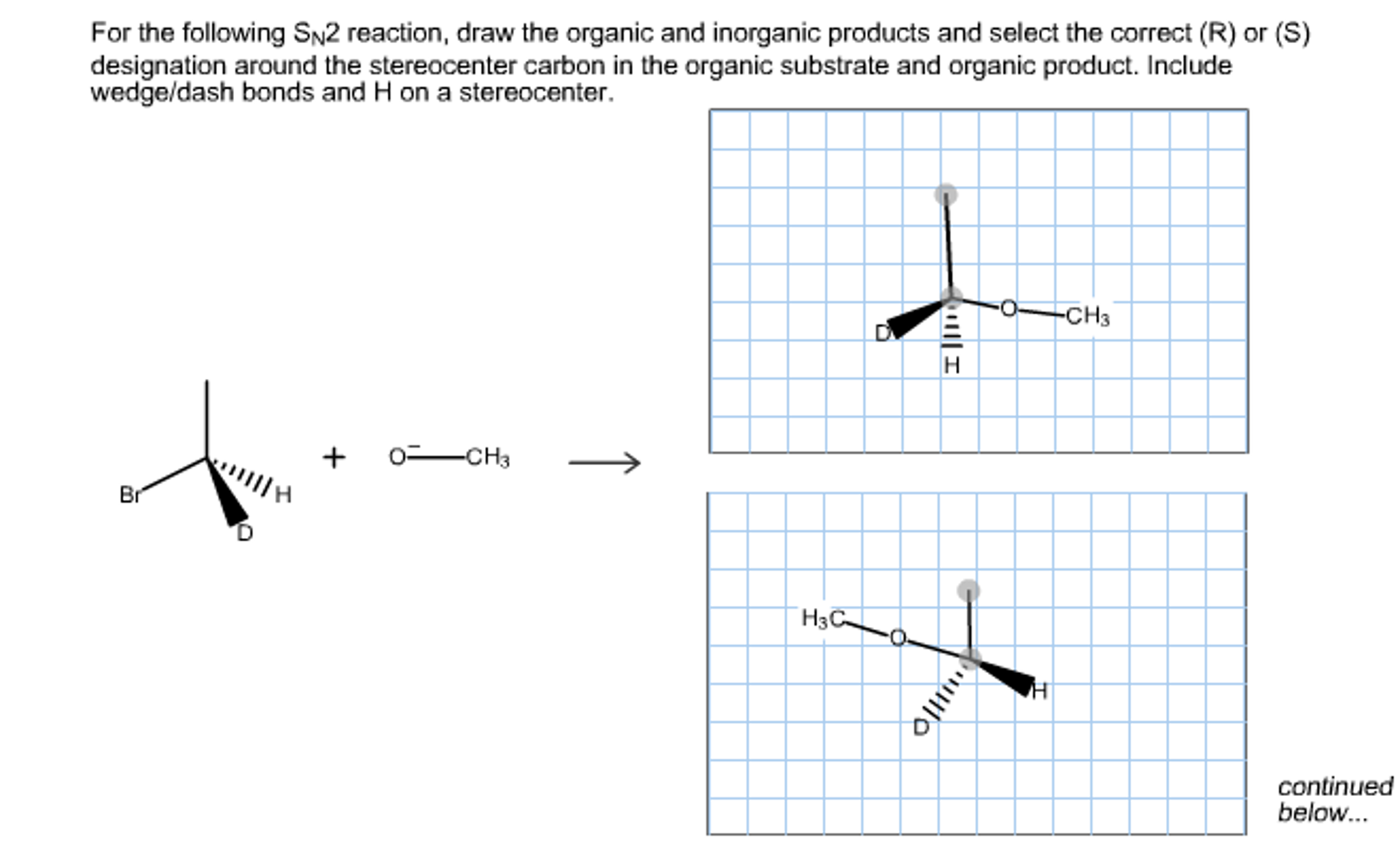
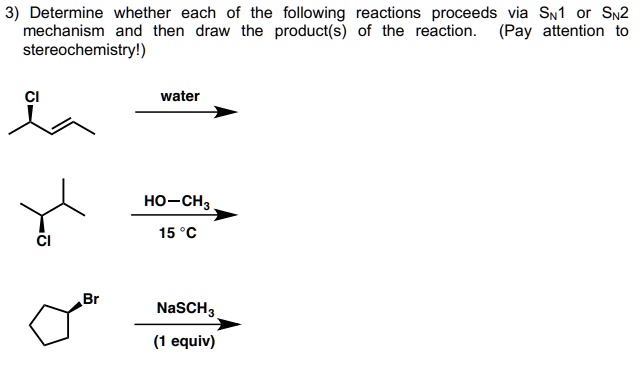
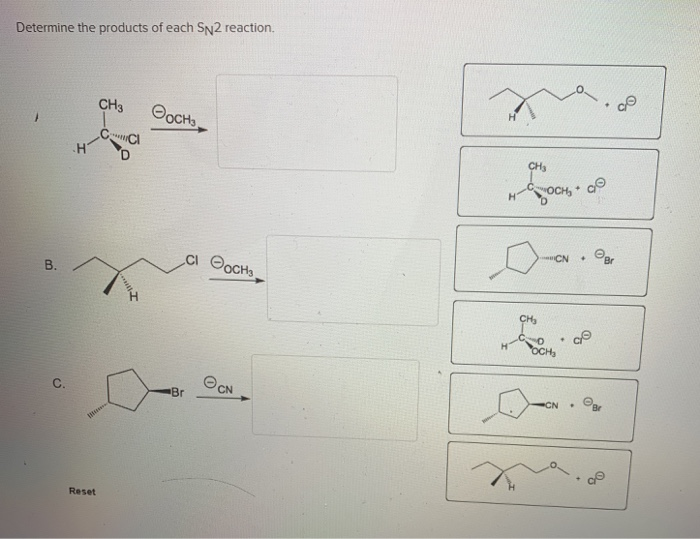
Determine The Products Of Each Sn2 Reaction.
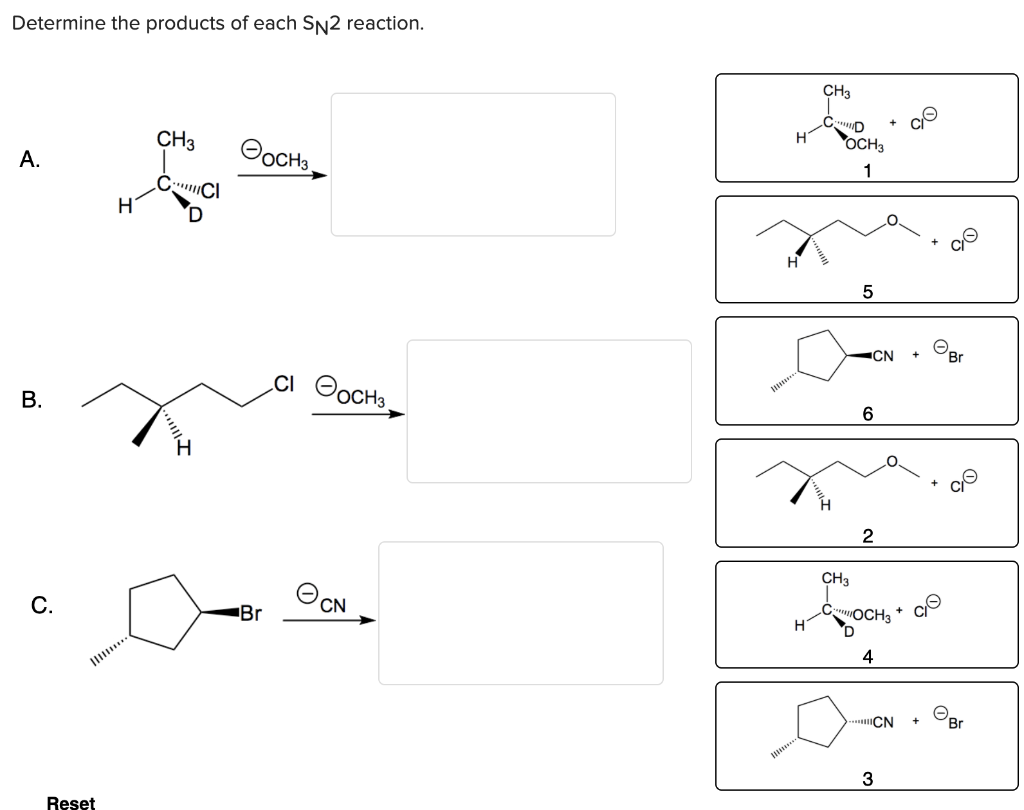
Hey there, my fellow chemistry adventurers! Grab your mug, settle in, and let's chat about something super cool: SN2 reactions. You know, those little molecular dance parties where one thing bumps another thing right out of the way? We're gonna figure out what pops out at the end of these things. No biggie, promise!
So, what's the deal with SN2? It's like a really polite but firm "excuse me" in the molecular world. SN2 stands for Substitution, Nucleophilic, Bimolecular. Fancy, right? But what it really means is that a nucleophile (that's the "pusher," the electron-rich attacker) comes in and attacks a substrate (that's the molecule being attacked). And bam! It kicks off a leaving group. Simple as that. Or… is it?
Let's break it down like we're dissecting a really complicated sandwich. We've got our main players. First up, the nucleophile. Think of it as the party guest who's really eager to get to the dance floor. It’s got extra electrons, ready to share (or, you know, attack). Common nucleophiles? Halides like Br⁻, I⁻, Cl⁻. Also, things like OH⁻ (hydroxide), CN⁻ (cyanide – ooh, spooky!), or even water molecules in a pinch. The stronger the nucleophile, the more likely it is to get this party started. It's all about electron density, folks!
Must Read
Then we have the substrate. This is the molecule that's about to get… rearranged. The key thing here is the carbon atom that the nucleophile is going to "visit." It needs to have a leaving group attached to it. What's a leaving group? Think of it as the current dance partner who's just not cutting it anymore. It’s gotta be something that can stabilize those extra electrons it’s gonna end up with. Good leaving groups are usually weak bases. Like halides again (Br, I, Cl). They're pretty chill about taking those electrons and peacing out. Tosylates and mesylates are also superstar leavers. They're like the VIPs of leaving groups, making the whole SN2 process a breeze.
Now, the "bimolecular" part of SN2? It means the rate of the reaction depends on the concentration of both the nucleophile and the substrate. It's a true collaboration! Both molecules have to be present for the magic to happen. It's not like the nucleophile just waits around for the substrate to do its thing alone. They gotta be in the same place at the same time. Like a perfectly timed duet. Or, you know, a collision.
The mechanism itself is pretty darn neat. It's a one-step process. No messy intermediates hanging around, no time for awkward pauses. The nucleophile comes in from the backside, opposite the leaving group. It's like a stealth attack! As the nucleophile starts to form a bond with the carbon, the bond between the carbon and the leaving group starts to break. It’s a delicate balance, a real push-and-pull. This transition state is where things get wild! Imagine a carbon atom sort of juggling five things for a split second – the incoming nucleophile, the outgoing leaving group, and the three other groups attached to the carbon. It's a fleeting moment of chaos, then…
Poof! The leaving group is gone, taking its electrons with it. And the nucleophile is now happily bonded to the carbon. The coolest part? This backside attack leads to inversion of stereochemistry. If the carbon atom was a chiral center, it flips completely inside out, like turning a glove inside out. So, if you started with an (R) configuration, you'll end up with an (S) configuration. Mind-blowing, right? It's like the molecule did a pirouette and ended up facing the other way.
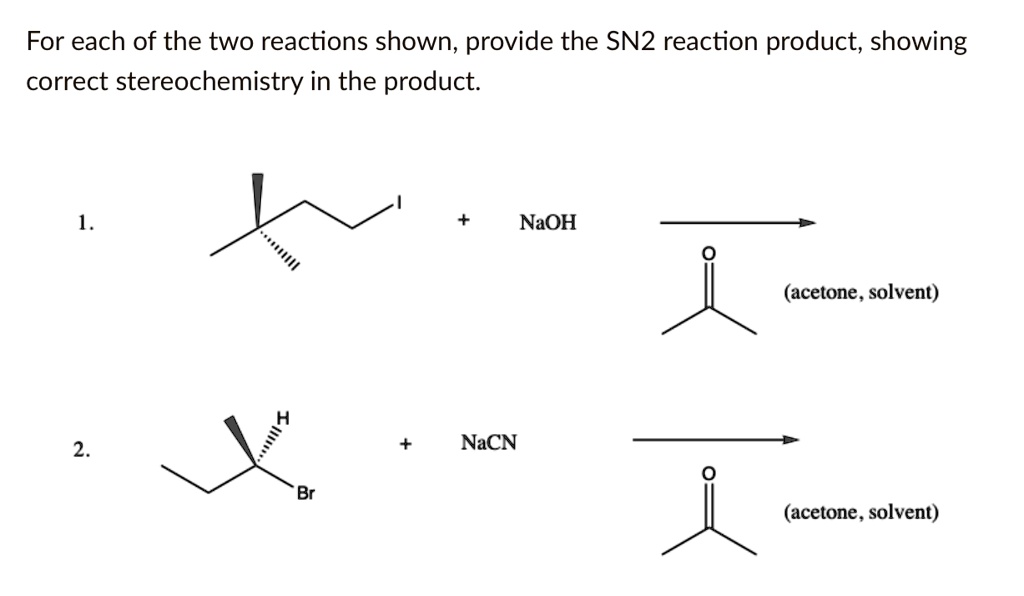
Okay, so how do we actually determine the products? It’s not rocket science, but you gotta pay attention. Think of it like this: you’re the detective, and the reaction is the crime scene. You need to identify the culprit (the nucleophile), the victim (the substrate and its leaving group), and then figure out who replaces the victim.
Step 1: Identify Your Players
First things first, look at your reaction. What's your nucleophile? Is it a strong electron-rich species? Is it negatively charged? Look for those clues! What's your substrate? You're looking for a carbon atom that's bonded to a leaving group. Remember those good leaving groups we talked about? Halides, tosylates, mesylates. If you don't see one of those, it might be an SN1 reaction or something else entirely. But for SN2, we need a good leaving group!
Let's do a quick example. Imagine you have CH₃Br (methyl bromide) and you're hitting it with OH⁻ (hydroxide). Your nucleophile is clearly OH⁻. Your substrate is CH₃Br. The carbon atom is bonded to Br, and bromide (Br⁻) is a decent leaving group. So, we're set for an SN2 reaction!
Step 2: The "Bump-Off" Strategy
This is where the actual product formation happens. The nucleophile, with its hungry electrons, is going to attack the carbon atom. And what's it going to replace? You guessed it, the leaving group. The nucleophile simply takes the spot of the leaving group. It's like musical chairs, but way more chemical.
So, in our CH₃Br + OH⁻ example: The OH⁻ will attack the carbon atom, and the Br⁻ will get kicked off. The new bond will be between the carbon and the oxygen from the hydroxide. So, you'll form CH₃OH (methanol). And the leaving group? It'll be Br⁻. So, your products are methanol and bromide ion.
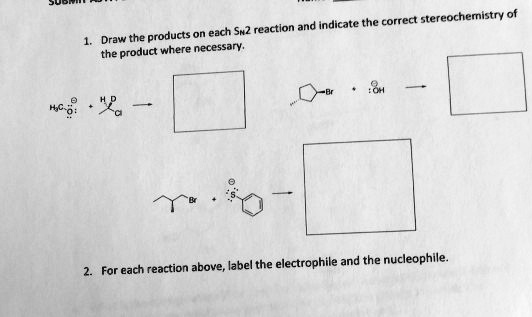
Step 3: Consider Steric Hindrance (The "Crowded Dance Floor" Problem)
This is a HUGE factor in SN2 reactions. Remember how the nucleophile has to attack from the backside? Well, if the carbon atom is all crowded up with bulky groups, it's going to be really hard for the nucleophile to get in there. Think of trying to do a fancy dance move when there are a bunch of people standing really close to you. Not fun!
SN2 reactions work best on primary (1°) and methyl substrates. These are carbons bonded to only one or zero other carbon atoms. They're like wide-open dance floors. Secondary (2°) substrates can also undergo SN2, but it's slower because there's more crowding. Tertiary (3°) substrates? Forget about it! They're way too crowded. The nucleophile just can't get to the backside. It's like a mosh pit in there! So, if your substrate is tertiary, an SN2 reaction is highly unlikely, and you should be looking for other reaction pathways, like SN1.
Let's say we have (CH₃)₃C-Br (tert-butyl bromide) and we try to react it with OH⁻. That carbon atom is attached to three methyl groups. That's a whole lot of bulk! The OH⁻ just can't sneak in. So, you're not going to get the SN2 product where OH replaces Br. It's more likely to go through an SN1 mechanism or elimination.
Step 4: Don't Forget the Stereochemistry! (The Glove Trick)
This is where things get really interesting, especially if your substrate has a chiral center. Remember the inversion of configuration? If you start with a chiral carbon with a specific stereochemistry (R or S), the product will have the opposite stereochemistry. This is a dead giveaway that you've got an SN2 reaction happening.
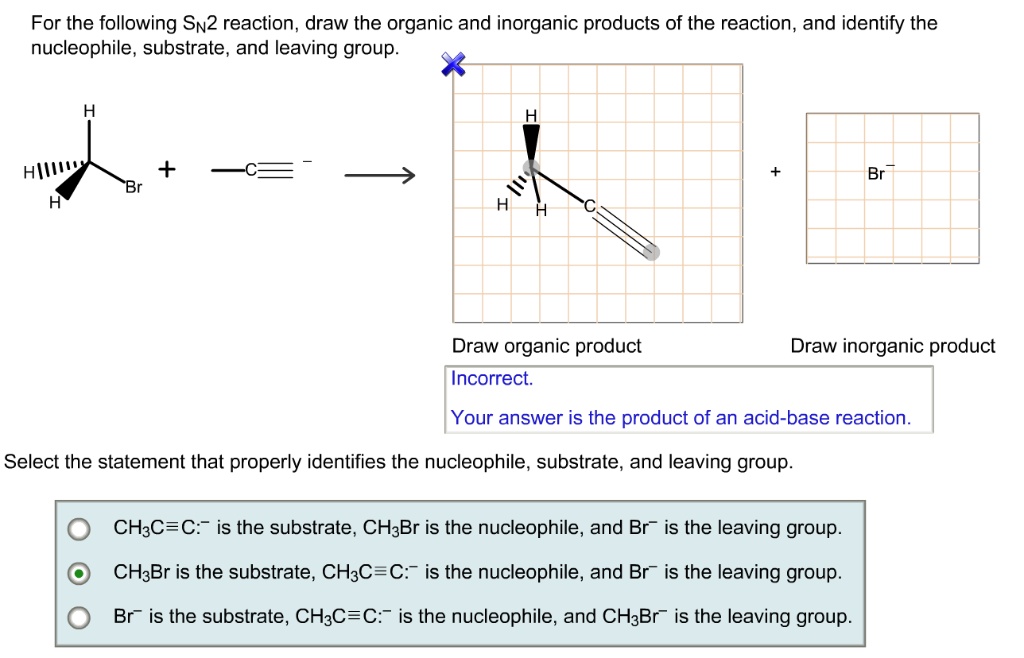
Let's imagine we have (R)-2-bromobutane reacting with CN⁻. The carbon at position 2 is chiral. Bromide (Br⁻) is our leaving group. Cyanide (CN⁻) is our nucleophile. Since it's a secondary substrate, SN2 is possible, especially with a good nucleophile like cyanide. The cyanide will attack from the backside, kicking off the bromide. If you started with (R)-2-bromobutane, you'll end up with (S)-2-cyanobutane. See? The configuration flipped! You've got to keep track of those R's and S's!
Step 5: What About the Solvent? (The Dance Floor Environment)
The solvent can play a role too, although it's a bit more subtle. SN2 reactions prefer polar aprotic solvents. What does that even mean? Well, "polar" means it has charged ends, like water. "Aprotic" means it doesn't have those easily removable hydrogen atoms that can hydrogen bond. Think of solvents like acetone, DMSO (dimethyl sulfoxide), or DMF (dimethylformamide).
Why do they like these solvents? Because polar aprotic solvents are good at dissolving the ions involved (like our nucleophile and leaving group), but they don't get in the way of the nucleophile's attack. They don't solvate (or surround) the nucleophile too tightly. In polar protic solvents (like water or alcohols), the solvent molecules can form a cage around the nucleophile, making it less reactive and slowing down the SN2 reaction. So, if your reaction is in acetone, you can be pretty confident SN2 is the main game in town.
Putting It All Together: A Quick Checklist
So, when you're faced with a reaction and asked to find the SN2 product, here's your mental flowchart:
- Is there a nucleophile? (Electron-rich, often negatively charged species.)
- Is there a substrate with a carbon attached to a leaving group? (Good leaving groups are key!)
- What's the degree of the carbon attached to the leaving group? (Methyl, 1°, 2° are best for SN2. 3° is a no-go.)
- If there's a chiral center, expect inversion of stereochemistry. (R becomes S, S becomes R.)
- What solvent is being used? (Polar aprotic solvents favor SN2.)
If all (or most) of these boxes are ticked, you're probably looking at an SN2 reaction. The product is formed by the nucleophile replacing the leaving group, with a backside attack and a flip in stereochemistry if applicable.
Let's try another one, just for kicks. Imagine reacting ethyl bromide (CH₃CH₂Br) with sodium cyanide (NaCN) in DMSO.
Nucleophile: CN⁻ (from NaCN). It's negatively charged and ready to rumble! Substrate: Ethyl bromide (CH₃CH₂Br). The carbon attached to Br is a 1° carbon. Leaving group: Bromide (Br⁻). A good leaver. Solvent: DMSO. This is a polar aprotic solvent, perfect for SN2!
All signs point to SN2! The cyanide will attack the carbon of the ethyl group, kicking off the bromide. So, the CN group will replace the Br. The product will be CH₃CH₂CN (propionitrile). The leaving group will be Br⁻.
Easy peasy, right? Well, mostly! Sometimes, competing reactions can happen. For instance, if you have a strong base as your nucleophile and a secondary or tertiary substrate, elimination (E2) can also occur. But for now, let's stick to our SN2 mission. The core idea is: nucleophile attacks, leaving group leaves, and usually, the stereochemistry flips.
So next time you see a reaction that looks like it's setting up for an SN2, just channel your inner molecular detective. Identify your players, understand the mechanics, and you'll be predicting those products like a seasoned pro. It's all about understanding the dance, the partners, and the environment. And with a little practice, you'll be a true SN2 master. Happy reacting!