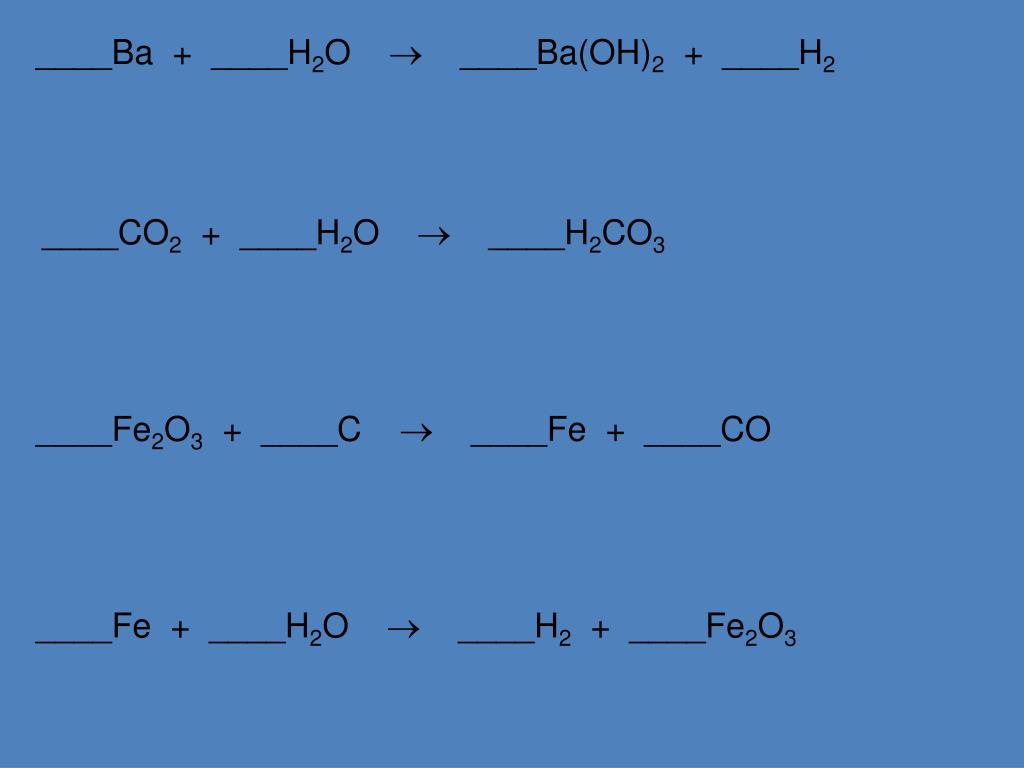
Balancing Chemical Equations Fe2o3 Co Fe Co2

Hey there, fellow adventurers in the wild and wonderful world of chemistry! Today, we're diving headfirst into a topic that might sound a tad intimidating at first glance, but trust me, it's about as exciting as finding a perfectly ripe avocado or a parking spot right in front of the grocery store on a Saturday. We're talking about balancing chemical equations!
Now, imagine you're at a party, and you've got a bunch of ingredients for a super-secret recipe. You've got iron(III) oxide, which looks like rusty-red dust, and carbon monoxide, which is a bit of a sneaky gas. These two fellas are about to get together and create something new, something exciting!
But here's the catch, my friends. In the grand theater of the universe, where atoms and molecules are the star performers, everything needs to be just so. It's like having a LEGO set – you can't just randomly stick bricks together and expect to build a magnificent castle, can you? Nope, you need to make sure all the pieces are accounted for!
Must Read
That's where our superhero skill of balancing chemical equations comes in. It's all about making sure that on one side of our chemical equation, we have the exact same "ingredients" as on the other side. Think of it as a cosmic bookkeeping system, ensuring no atom is left behind or suddenly appears out of thin air. It's a rule of nature, as fundamental as gravity making sure your toast lands butter-side up (or down, depending on your luck!).
Let's peek at our specific equation today: Fe2O3 + CO → Fe + CO2. This is like our starting point, our initial guest list for the chemical party. On the left, we have our lovely iron(III) oxide (Fe2O3) and our wily carbon monoxide (CO). They're the guests who arrived first, ready to mingle.
On the right side, we have the results of their epic chemical romance: pure iron (Fe), looking all shiny and metallic, and carbon dioxide (CO2), the gas we exhale when we're feeling particularly enthusiastic. But, as you can see with your eagle eyes, the numbers just aren't matching up yet! It's like showing up to a party with 5 cupcakes but only having 3 plates – a culinary crisis in the making!

So, our mission, should we choose to accept it (and we totally should because it's awesome!), is to add some coefficients. These are like little numbers we put in front of our chemical formulas. They don't change what the molecules are, just how many of them are participating in the reaction. It's like deciding how many appetizers to serve, not changing the appetizer itself from mini quiches to sushi.
Let's start by looking at our iron (Fe). On the left side of our equation, we have two iron atoms tucked away inside that Fe2O3. But on the right side, we only have one lonely iron atom. That's like having two movie tickets but only one person to give them to – not very equitable!
To fix this, we need to tell ourselves, "Okay, we need two of those iron atoms on the right side." So, we magically place a big '2' in front of our Fe. Our equation now looks like this: Fe2O3 + CO → 2Fe + CO2. See? We're already making progress! It's like adding an extra friend to the dance floor!
Now, let's turn our attention to the oxygen atoms. This is where things get a little more intricate, but don't let that deter you! We have three oxygen atoms in Fe2O3 and one in CO on the left, for a total of four. On the right, we have two oxygen atoms in CO2. We're still a bit short on the right side, or perhaps the left side has too many. It's a classic "who has more cookies?" scenario!
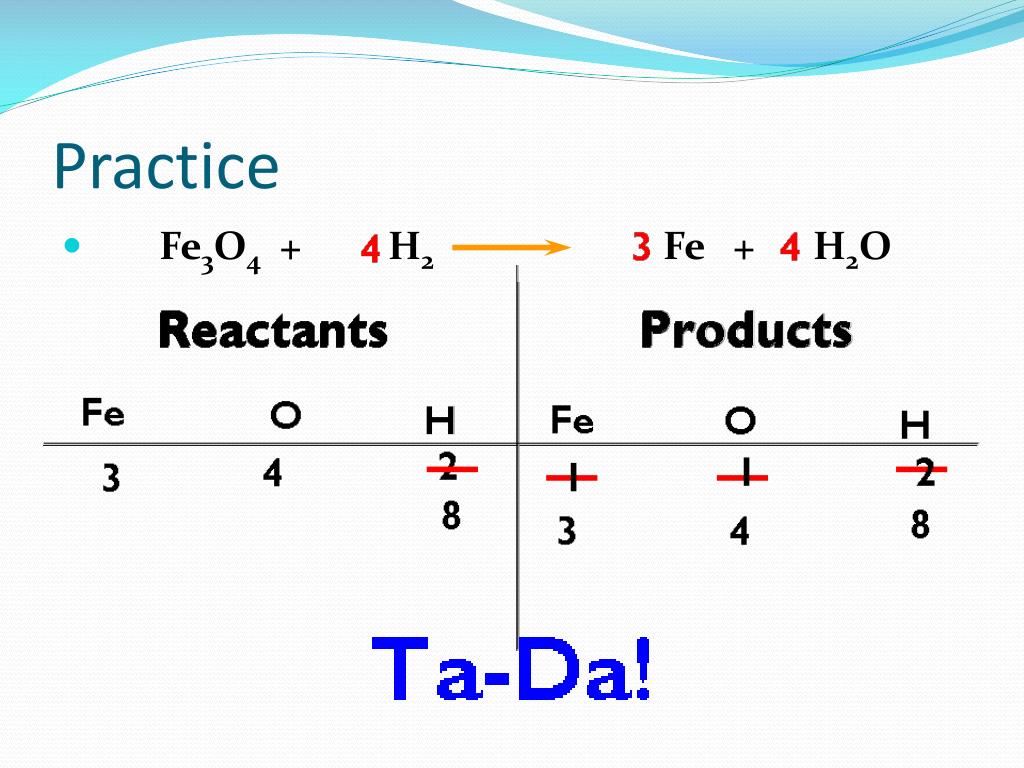
This is where the carbon monoxide (CO) and carbon dioxide (CO2) come into play. They're like the tag-team champions of our equation. If we put a '2' in front of CO on the left, we get Fe2O3 + 2CO → 2Fe + CO2. Now we have 2 carbons and a total of 3 + 2 = 5 oxygen atoms on the left. Still not quite there!
Let's try a different approach with our oxygen. We need more oxygen on the right side to balance out what's on the left. Remember, we have 3 oxygens in Fe2O3 and we're thinking about how many CO molecules we need to react. What if we aim for a common ground for our oxygen atoms?
Let's go back to the iron. We've got our 2Fe on the right, which is solid. Now, let's think about the oxygen. We have 3 oxygens in Fe2O3. Our CO on the left needs to contribute some oxygen, and our CO2 on the right needs to take some away. It's a molecular exchange program!
Consider this: what if we had *three molecules of carbon monoxide (CO) on the left? That would give us a total of 3 + 3 = 6 oxygen atoms on the left. Our equation would look like: Fe2O3 + 3CO → 2Fe + CO2. Now we have the iron balanced, but the oxygen is still a bit wobbly. We have 6 oxygens on the left and only 2 on the right!
This means we need more CO2 on the right to soak up all that extra oxygen. If we have 6 oxygens on the left from Fe2O3 and 3CO, we need to make sure the right side also has 6 oxygens. Since each CO2 has 2 oxygens, we'd need three molecules of CO2 to get us to 6 oxygens!
So, let's try this: Fe2O3 + 3CO → 2Fe + 3CO2. Let's do a final check, shall we? On the left: two iron atoms, three + (3 * 1) = 6 oxygen atoms, and 3 carbon atoms. On the right: two iron atoms, (3 * 2) = 6 oxygen atoms, and 3 carbon atoms. Ta-da! It's a perfect match!
We have successfully balanced our equation! It's like making sure every dancer in the ballet has a partner and the costume department has the exact number of sequins. Everything is in its rightful place, and the chemical performance can go off without a hitch.

This process, my friends, is the magic of balancing chemical equations. It's not just about numbers; it's about respecting the fundamental laws of the universe. It's about ensuring that matter is conserved, meaning it can't be created or destroyed, only rearranged. Pretty neat, huh?
So, the next time you see a chemical equation, don't shy away. Think of it as a puzzle, a fun brain teaser waiting to be solved. With a little practice and a dash of enthusiasm, you too can become a master balancer of all things chemical. It's a skill that will serve you well, whether you're a budding chemist or just someone who appreciates the elegant order of the world around us.
Remember, iron(III) oxide and carbon monoxide, when properly balanced, transform into the glorious iron and the ever-present carbon dioxide. It's a beautiful dance of atoms, orchestrated by the principles of conservation. So go forth, my friends, and balance with glee!

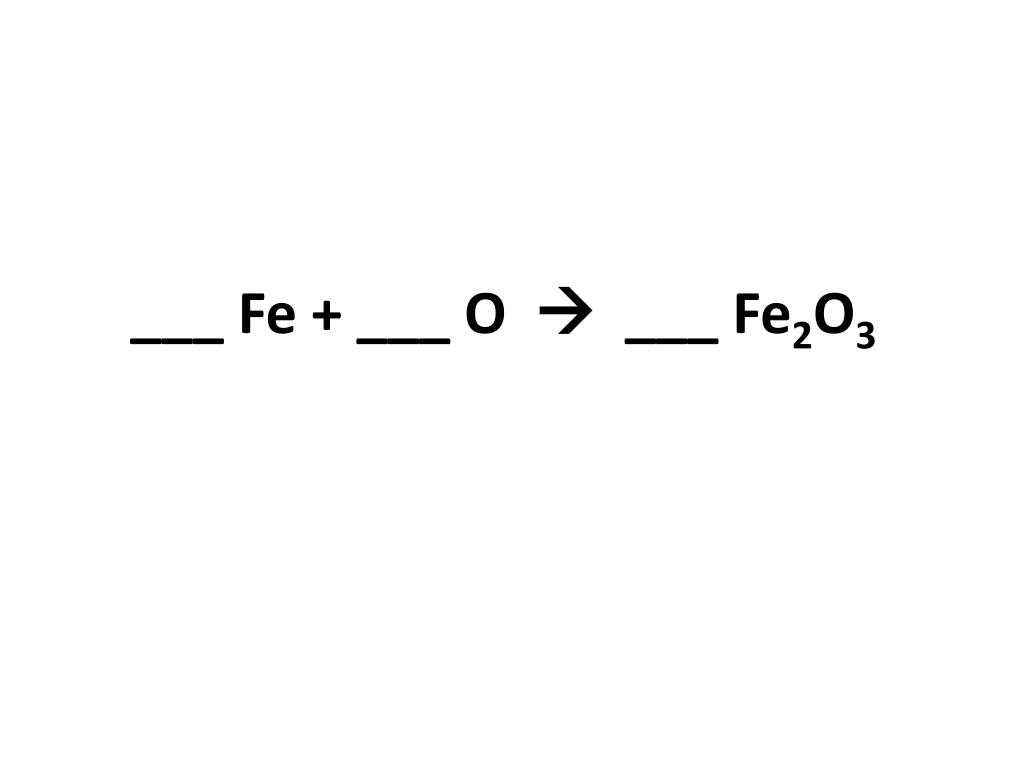
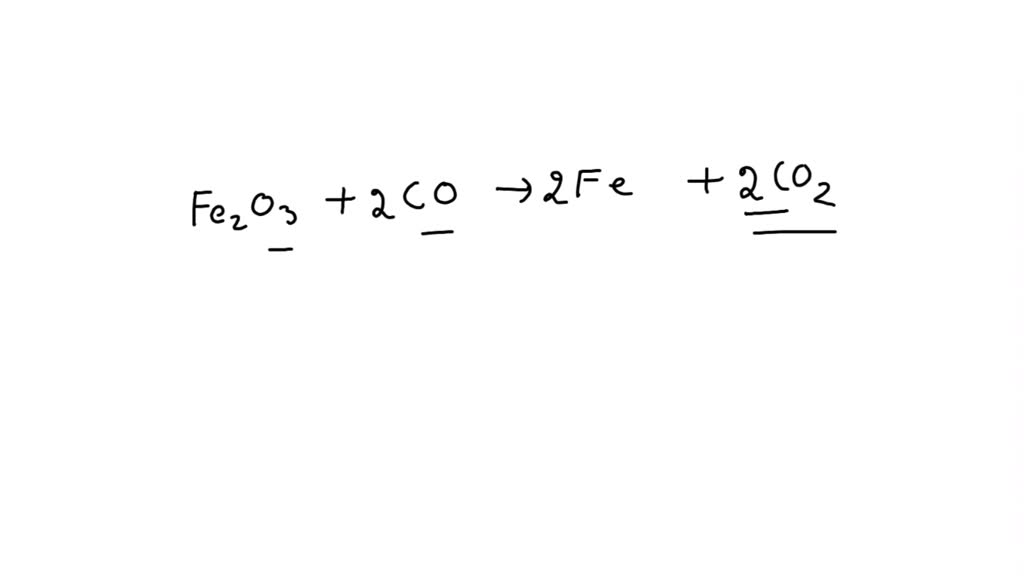
![[ANSWERED] Read the chemical equation Fe2O3 CO Fe CO If 2 moles of Fe](https://media.kunduz.com/media/sug-question-candidate/20220509163846738247-4555698.jpg?h=512)